Clinical Skills
Discover guidance for assessing patients, diagnostic testing, and surgical procedures through expert-led articles and videos. Perfect for general practitioners, veterinary students, and residents looking to refine their skills.
A young dog was presented with painful, rapidly progressive vesicles and ulcers. Review the diagnostic steps that confirmed epidermolysis bullosa acquisita and the immunosuppressive strategy that led to remission.
Topics In Clinical Skills
Featured in Clinical Skills
Poll
Brought to You by Nutramax

Quiz
Brought to You by Nutramax

New in Clinical Skills
Sponsored byHill's Pet Nutrition
Sponsored byNutramax Laboratories
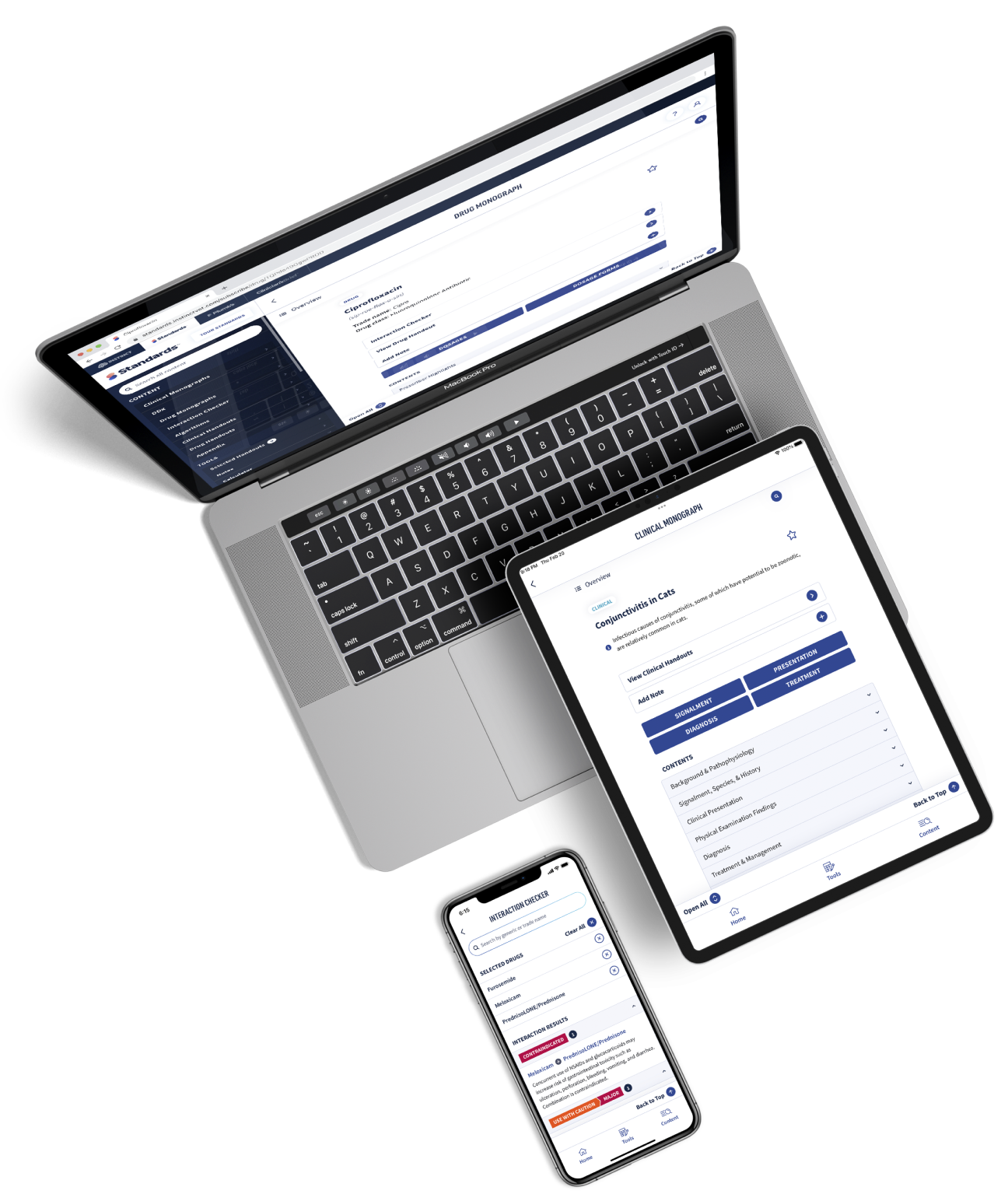
Get more clinical guidance with Standards of Care™
From the team that brings you Clinician’s Brief, extend your knowledge with expert-written, peer-reviewed diagnostic and treatment guidance as well as pet owner education and all the reliable drug information you love in Plumb’s.