Clinical Skills
Discover guidance for assessing patients, diagnostic testing, and surgical procedures through expert-led articles and videos. Perfect for general practitioners, veterinary students, and residents looking to refine their skills.
Haws syndrome is an often self-limiting condition in cats causing bilateral third eyelid elevation that is often accompanied by diarrhea. This recent study looked at 5 cases to better understand the condition’s clinical features, outcomes, and potential treatments.
Topics In Clinical Skills
Featured in Clinical Skills


New in Clinical Skills
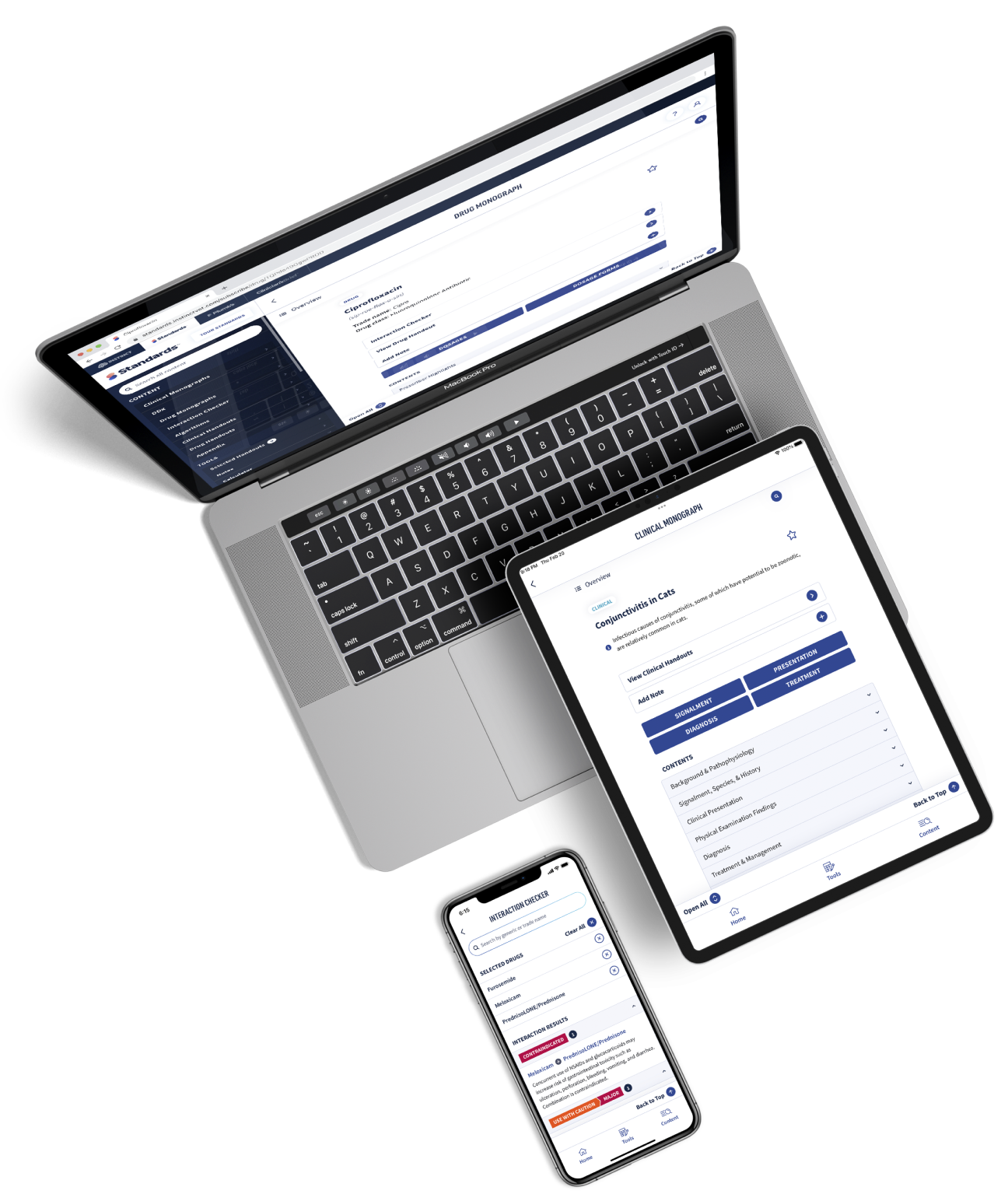
Get more clinical guidance with Standards of Care™
From the team that brings you Clinician’s Brief, extend your knowledge with expert-written, peer-reviewed diagnostic and treatment guidance as well as pet owner education and all the reliable drug information you love in Plumb’s.