Drugs & Therapeutics
Learn about the latest veterinary medications and treatment options, including dosages, side effects, and interactions, from experts in the field.
Sponsored By 

For appropriate candidates, SGLT2 inhibitors offer effective glycemic control without the compliance challenges of insulin therapy. Get up to speed on the indications, risks, and treatment goals for this growing treatment option in feline diabetes mellitus.
Topics In Drugs & Therapeutics
Featured in Drugs & Therapeutics
New in Drugs & Therapeutics
31m:52s
Ep. 376
Sponsored byHilltop Bio
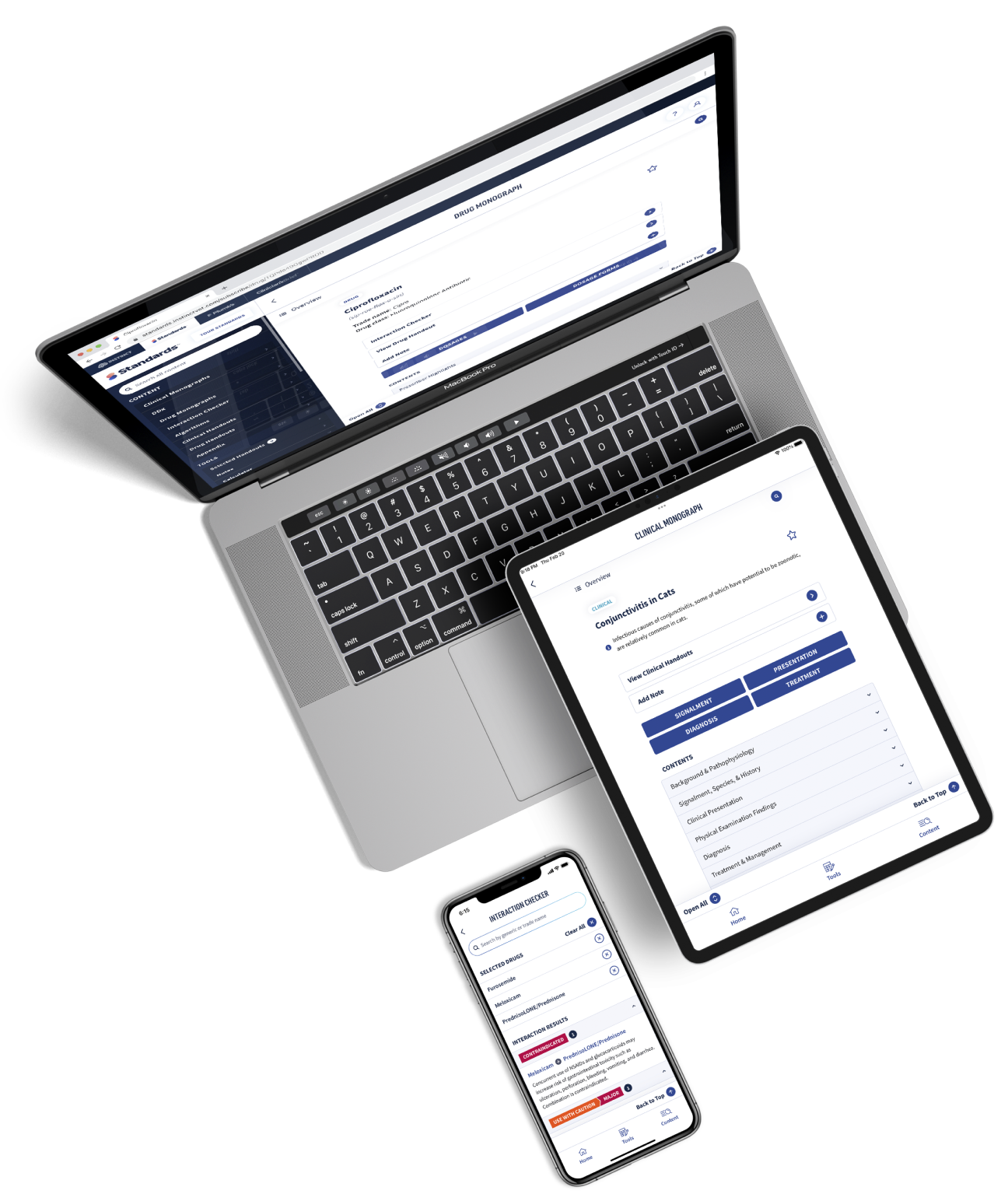
Get more clinical guidance with Standards of Care™
From the team that brings you Clinician’s Brief, extend your knowledge with expert-written, peer-reviewed diagnostic and treatment guidance as well as pet owner education and all the reliable drug information you love in Plumb’s.