Top 5 Liver Conditions in Dogs
Craig B. Webb, PhD, DVM, DACVIM (Small Animal), Colorado State University
The liver serves as the control-and-command center for virtually all metabolic processes: production, packaging, and distribution of proteins, lipids, and carbohydrates; hormonal and enzymatic control of metabolic pathways; metabolism of biologics; transformation of xenobiotics; decontamination and removal of toxins; and recirculation, recycling, and refilling of gallbladder contents. When the liver does not function at full capacity, clinical manifestations are often ubiquitous and possibly devastating.
TOP 5 Liver Conditions in Dogs
Hepatitis or hepatic insult
Hepatic fibrosis or cirrhosis
Copper-associated hepatitis
Congenital portosystemic vascular anomalies (PSVA)
“Nonhepatic” hepatic disease
1. Hepatitis or hepatic insult
Acute
The liver can be exposed to ingested toxins, blood-borne pathogens, and drugs and their metabolites, providing numerous causes for acute insult. Signs can include anorexia, fever, vomiting, and abnormal mentation. Jaundice is the classic sign of hepatic failure, with RBC hemolysis as another important differential for hyperbilirubinemia.
Leptospirosis appears to be increasingly prevalent and usually involves both the liver and kidneys. Other possible infectious agents include infectious canine hepatitis (canine adenovirus-1), Clostridium piliformis, bacteria (especially Escherichia coli), and Toxoplasma gondii. Ingestion of Amanitum spp mushrooms, blue–green algae, and some drugs (eg, sulfonamides, carprofen, amiodarone) can result in acute and significant liver disease. Consequences of insult may be idiosyncratic and unpredictable, and signs may vary.
ALT and bilirubin are the most pertinent biochemical indicators of hepatic insult, although low BUN, albumin, cholesterol, and glucose levels along with prolonged clotting times are indicative of fulminant hepatic failure. Supportive care is essential. Glucocorticoids may be contraindicated (infection) or of minimal benefit in the acute setting. Acetylcysteine has been used in critical patients with a loading dose of 140 mg/kg IV, followed by additional treatments at 70 mg/kg.
Related Article: Top 5 Liver Conditions in Cats
Related Article: Chronic Liver Disease
Chronic
Signs of chronic canine hepatitis, often nonspecific and systemic, include vomiting, lethargy, decreased appetite, polyuria/polydipsia (PU/PD), and weight loss. Increased ALT enzyme activity, usually the most telling biochemical abnormality, is frequently monitored as a quantifiable indicator of treatment response.
Although the primary disease cause may be undetermined, several treatable causes warrant diagnostic examination with histopathology, metal analysis, and bacterial culture. Chronic hepatitis may present as a slow progression of changes started by acute insult. Culturing bile, not liver tissue, may yield better growth.1 There is often an immune-mediated component to chronic hepatitis progression, warranting immunomodulatory therapy. Prednisone has been the preferred drug, but Colorado State University has recently had success using cyclosporine at a starting dose of 5 mg/kg q24h.2 Unlike prednisone, cyclosporine does not induce canine liver enzyme elevation.
Hepatitis cases (acute and chronic) can be treated with ursodeoxycholic acid at 7.5 mg/kg q12h to enhance bile flow, dilute toxic bile acids, and provide both immunomodulatory and antioxidant effects. Antioxidants may benefit cases of canine hepatitis: S-adenosylmethionine (SAMe; 10 mg/kg q12h), silymarin (100–200 mg/dog q24h), and vitamin E (100–400 IU/day).
PSVA = portosystemic vascular anomalies, PU/PD = polyuria/polydipsia
2. Hepatic fibrosis or cirrhosis
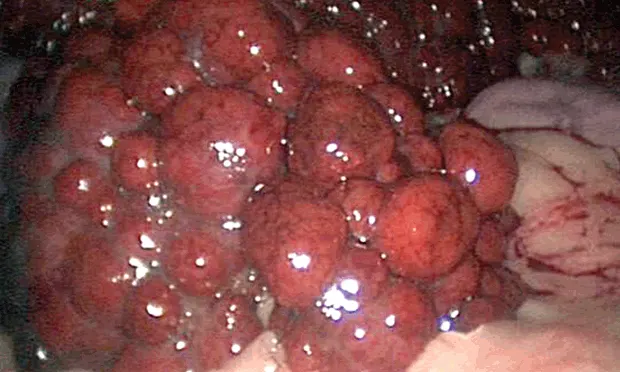
Chronic hepatitis may progress to a cirrhotic liver (Figure 1) as fibrin replaces liver parenchyma, causing permanent changes in hepatic architecture. This is a morphologic diagnosis of prognostic significance. The presentation, often severe, can include weight loss, ascites, and jaundice. A PCV/TP can quickly rule out hemolytic anemia as the cause of jaundice, and a serum bile acids test is redundant if the total bilirubin is significantly elevated. Ascites may be a result of portal hypertension from the cirrhotic liver, hypoalbuminemia from loss of liver function, retention of sodium or water via stimulation of the renin–angiotensin–aldosterone system, or a combination thereof.
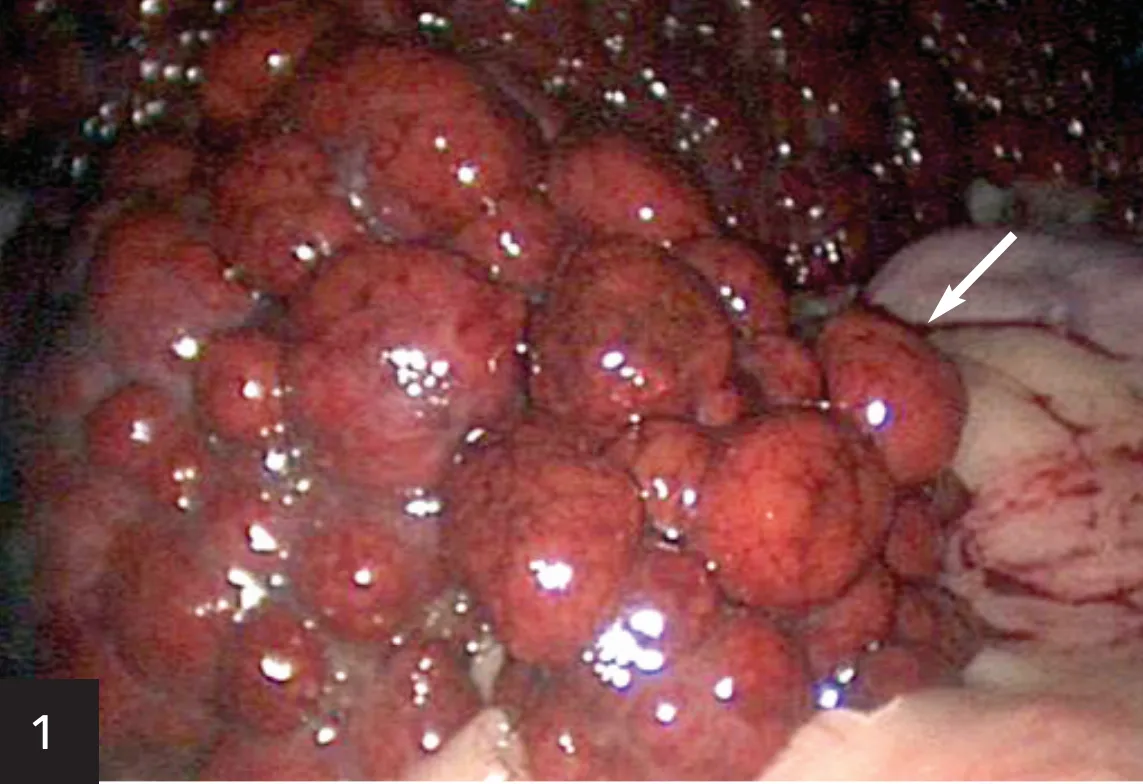
Diffuse, severe nodular changes (arrow) throughout all lobes of the liver and a decrease in liver size, consistent with hepatic cirrhosis. As hepatic parenchyma is lost and scar tissue forms, the liver attempts to regenerate, but all semblance of normal architecture (and eventually function) is lost. One alternative rule-out might be benign hepatic nodular hyperplasia, but case presentation and biochemical abnormalities would distinguish the two differentials before biopsy (moderate elevation in ALP activity as compared to systemic signs of liver failure).
Although the diagnostic work-up (eg, histopathology, metal analysis, culture) is similar to that of less marked cases of chronic hepatitis, it is often too late for specific beneficial treatment, even if a primary cause is identified (eg, copper-associated hepatitis). Supportive care may include palliative abdominocentesis if the ascites compromises respiration, but the newly emptied space will likely refill. Spironolactone (1–2 mg/kg q24h) combined with furosemide may be used when fluid removal is less critical; however, it may increase risk for hepatic encephalopathy from alkalosis or hypokalemia effects. Cirrhosis and ascites are negative prognostic indicators against which colchicine at 0.025 mg/kg q24h may be tried; however it lacks proven benefit. Prednisone and nonspecific liver protectants are indicated, but the prognosis can be grave.
3. Copper-associated hepatitis
As a form of chronic hepatitis, copper-associated hepatitis is thought to result from an inherited enzymatic defect in several breeds, especially Bedlington terriers but also Dalmatians, Labrador retrievers, and West Highland white and Skye terriers. Excessive hepatic copper is a treatable component of chronic hepatitis cases (common in Doberman pinschers, cocker spaniels, mixed breeds) in which copper accumulation may be secondary to the disease. The copper chelator d-penicillamine at 10 to 15 mg/kg q12h is preferred for removing excess copper, and elemental zinc at 10 mg/kg q12h can decrease GI copper absorption; however, the two should not be used simultaneously.
Most commonly seen as a chronic hepatopathy, copper-associated liver disease can present as an acute illness or acute hemolytic crisis (rare), so qualitative or quantitative assessment of hepatic copper content should be a standard diagnostic of liver biopsy.
Related Article: Increased Liver Enzyme Activity in a Dog
4. Congenital portosystemic vascular anomalies (PSVA)
The liver is normally the first stop for blood carrying digestive and absorptive efforts of the GI tract to the rest of the body. Various congenital vascular anomalies can result in blood bypassing the liver, causing release of digestive content systemically without screening or preparation and resulting in decreased liver function. The nomenclature has recently been clarified by WSAVA Standards.
The most common presentation is in young dogs with stunted growth and/or abnormal mentation, but more subtle signs of PSVA (eg, GI, PU/PD, weight loss, lethargy, poor hair coat) in older pets are increasingly appreciated; other signs are generally related to the nervous or urinary system. Serum biochemistry profile and urinalysis may reflect the dysfunction (ie, low or low-normal glucose, cholesterol, albumin, BUN, microcytic anemia or target cells, ammonium biurate crystalluria); however, pre- and postprandial serum bile acids testing (not liver enzyme elevation) may best establish decreased liver function, although it is not specific to PSVA.
When the clinical presentation and serum bile acids test are consistent with decreased liver function, PSVA can be confirmed and characterized by abdominal ultrasonography. A single extrahepatic (small breed) or intrahepatic (large breed) shunt is frequently visualized. Advanced imaging or histopathology can be used if ultrasonography is equivocal or if primary portal vein hypoplasia (formerly microvascular dysplasia or noncirrhotic portal hypertension) is suspected.
It is necessary to determine whether the PSVA patient requires surgical (most single shunts) or nonsurgical (primary hypoplasia, multiple acquired shunts) treatment. Medical management includes dietary intervention (low-quantity, high-quality protein) and pharmaceutical manipulation of gut ammonia production (lactulose, 0.5 mL/kg q8–12h; neomycin, 22 mg/kg q8h).
5. “Nonhepatic” hepatic disease
Nonhepatic liver disease includes the following reactive hepatopathies: vacuolar hepatopathy, steroid hepatopathy, and benign nodular hyperplasia. Elevations in liver enzyme activities do not necessarily indicate primary liver disease. The liver senses various conditions or diseases and reacts with enzyme elevations; hence the term reactive hepatopathy. Overlooking these differentials may lead to unproductive diagnostic efforts.
Steroids and anticonvulsants can induce elevated canine liver enzyme activities. Benign nodular hyperplasia in older dogs can mimic enzymatic and ultrasonographic changes similar to those seen in neoplastic disease. Growth in young dogs, bone cancer in older dogs, and endocrinopathies (eg, Cushing’s disease, diabetes mellitus) can result in elevated liver enzymes. Idiopathic vacuolar hepatopathy is a histopathologic diagnosis consistent with exogenous steroid administration, Cushing’s disease, or other systemic illness. After identifying and treating the primary disease, nonspecific therapy (eg, antioxidants) aimed at the liver may be administered. Elevated liver enzymes in a dog without clinical signs may be reevaluated in 4 to 6 weeks before pursuing a more extensive diagnostic work-up.
Closing thoughts
The liver is involved in most every aspect of life. Therefore, primary liver disease makes the list of differentials for most presentations, while secondary liver involvement must be considered in most nonliver diseases.