Intestinal surgery is frequently performed in veterinary medicine and may be indicated for various reasons, including obstructive foreign bodies, trauma, biopsies, or neoplasia. Postoperative complications following intestinal surgery in dogs and cats can lead to significant morbidity and mortality. The most significant and common complications include intestinal dehiscence, postoperative ileus (POI), surgical site infection, and delayed return to eating, all of which can severely impact recovery and survival.
This article reviews the pathophysiology, diagnosis, treatment, and prevention strategies for several common complications.
1. Intestinal Dehiscence
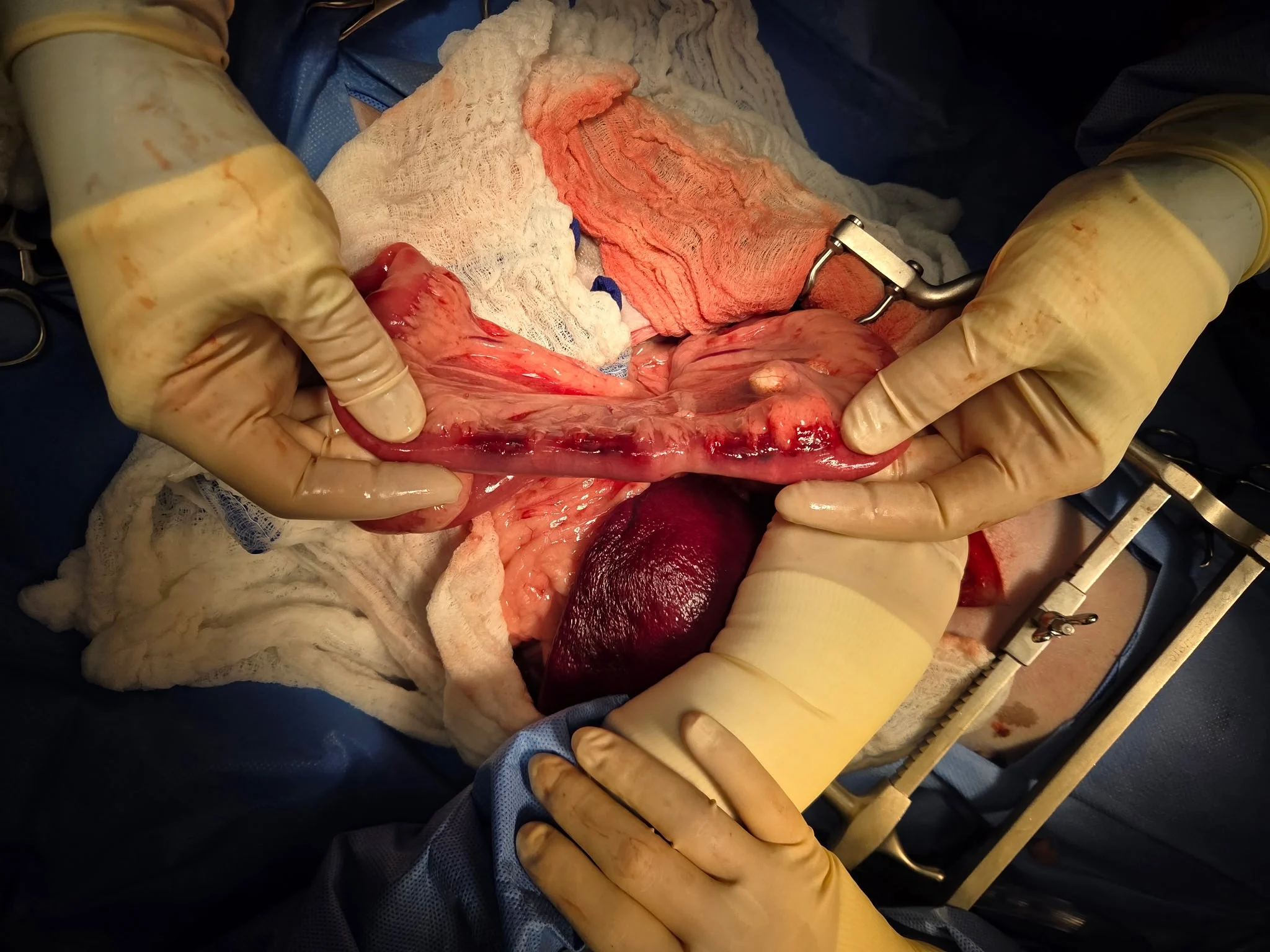
Surgical dehiscence is the full or partial opening of a surgical site postoperatively. Dehiscence of the intestinal surgical site is a serious complication that can result in potentially fatal septic peritonitis secondary to leakage of intestinal contents into the abdomen. This complication occurs most often during the inflammatory phase of healing when the biomechanical (tissue) strength of anastomosis is critically low and heavily reliant on the integrity of the anastomotic (suture or staple) technique.1 Some factors (eg, preoperative peritonitis/inflammation, hypoalbuminemia, intraoperative hypotension, presence of a foreign body) can predispose patients to this complication (Figure 1).2 Intestinal dehiscence usually occurs within 2 to 5 days after surgery, and clinical signs typically include depression, anorexia, vomiting, and abdominal pain. The overall rate of dehiscence in dogs ranges from 3% to 28%, with mortality rates up to 85%.1,3-6 Risk factors for postoperative septic peritonitis after intestinal surgery include a pre-existing septic abdomen, preoperative hypoalbuminemia, preoperative hypoproteinemia, and intraoperative hypotension.7 Mortality associated with septic peritonitis has been linked to several risk factors, including hepatic injury, acute kidney injury, prolonged prothrombin time, and intraoperative or postoperative hypotension.8


FIGURE 1 Jejunum after foreign body removal, with bruising noted along the mesenteric side of the intestine
Peritoneal effusion may be present in cases of intestinal dehiscence with suspected septic peritonitis; however, absence of peritoneal effusion does not rule out intestinal dehiscence, as focal or localized peritonitis may occur without significant fluid accumulation.
Abdominocentesis should be performed to evaluate the fluid further. Glucose and lactate levels can be measured in the peritoneal fluid and compared to peripheral blood values; a fluid-to-blood glucose difference of >20 mg/dL and a lactate gradient of >2 mmol/L indicate sepsis.9 The fluid should also undergo cytologic analysis for evidence of infection. Septic fluid typically contains a high number of neutrophils (with many showing degenerative or toxic changes), a small number of macrophages, and intracellular bacteria; however, absence of intracellular bacteria does not rule out septic peritonitis. The presence of bacteria may be low within the abdominal fluid, and culture is warranted for further evaluation.
If available, postoperative ultrasonography may be performed 48 to 96 hours following intestinal surgery to enable early and sensitive detection of anastomotic integrity and the potential absence of dehiscence.10 Abdominal ultrasonography may be warranted earlier if clinical signs of dehiscence are seen. Characteristic ultrasonographic findings associated with dehiscence include loop corrugation, intramural gas bubbles, steatitis, visible discontinuity of the intestinal wall, localized effusion, ileus, and wall thickening.10 Intramural gas and localized effusion are considered the most reliable indicators of intestinal leakage.10 Conversely, absence of steatitis and local effusion has been suggested as a favorable prognostic indicator, predicting an uncomplicated postoperative course.10
Several studies have compared the incidence of dehiscence between handsewn and stapled anastomoses. Stapled anastomoses were associated with a significantly lower dehiscence rate in dogs with preoperative peritonitis compared to handsewn techniques (9.7% vs 28.9%, respectively) in one study.3 Another study showed no difference in dehiscence frequency when comparing stapled anastomoses to handsewn anastomoses.11
Gentle tissue handling, appropriate suture selection, and adequate blood supply to the intestinal segments are optimal to minimize risk for surgical dehiscence. Intraoperative assessment for viability and tension-free closure is critical. Postoperative care should involve appropriate analgesia, early nutritional support, and close monitoring for signs of sepsis.
2. Postoperative Ileus
POI is a common and clinically significant complication following abdominal surgery that may contribute to significant postoperative morbidity, including delayed enteral nutrition, patient discomfort, and prolonged hospitalization.12 Clinical signs of POI may include anorexia, abdominal distension, vomiting, and decreased fecal output.13 Following surgery, GI motility is temporarily impaired, characterized by uncoordinated electrical activity and lack of effective propulsive movements.12 Small intestinal motility typically recovers within a few hours,14 gastric function generally normalizes within 24 to 48 hours,15 and the colon usually normalizes after 48 to 72 hours.14
Autonomic nervous dysfunction appears to be the primary initiator of POI. The sympathetic nervous system, which is generally inhibitory to the GI tract, becomes hyperactive in the postoperative period, causing a decreased release of acetylcholine and increased inhibition of motility.13 Risk factors include prolonged surgical time, peritonitis, extensive bowel handling, and inadequate analgesia.13 Ultrasonographic findings in patients with POI may include dilated, fluid-filled intestinal loops with reduced or absent peristalsis.16
Prokinetic agents (eg, metoclopramide, cisapride, erythromycin, ranitidine) may be used to enhance GI motility; however, erythromycin may not constitute judicious antimicrobial use, even at subantimicrobial dosages.17 Nasogastric tubes may be indicated in cases of ileus to decompress the stomach, prevent gastric distention, and reduce risk for aspiration due to regurgitation. Removal of accumulated gastric fluid via the tube can provide relief of clinical signs and help monitor GI function. Laparoscopic surgeries consistently reduce duration of POI compared to open procedures.12,18 Early ambulation and minimization of opioid use through multimodal analgesia strategies have also been associated with reduced duration and severity of POI.13 Prompt recognition and appropriate management are essential to improving postoperative recovery and minimizing morbidity in affected patients.
3. Surgical Site Infection
Clean surgery involves nontraumatic, noninfected procedures without entry into the respiratory, GI, genitourinary, or oropharyngeal tracts (eg, elective orthopedic procedures). Clean-contaminated surgery involves controlled entry into these tracts without significant contamination (eg, enterotomy in a stable patient). Contaminated surgery includes open, fresh, or traumatic wounds; major breaks in aseptic technique; or gross spillage from the GI tract. Dirty surgery involves the presence of infection, devitalized tissue, or perforated viscera (eg, septic peritonitis from intestinal rupture).19
Intestinal surgery is most commonly classified as clean-contaminated surgery and is reclassified as dirty surgery in the presence of septic peritonitis. Clean-contaminated surgical site infection rates in veterinary medicine range between 4.5% and 5%.18,20A study focusing on the rate of incisional infections after GI surgery in dogs and cats noted an overall incisional infection rate of 7%.21 Risk factors for surgical site infections include length of anesthesia, length of surgery, and incidence of underlying disease.20 Surgical site infections can lead to prolonged hospitalization and increased morbidity and mortality rates.21,22 Clinical signs include local pain, fever, purulent discharge, or abscessation.23
Escherichia coli and Staphylococcus pseudintermedius are commonly isolated from incisional infections after intestinal surgery.20-22 A study investigating incisional infections following GI surgery in dogs and cats reported that cefazolin and cefoxitin were the most commonly used perioperative antibiotics21; however, only 2 isolated bacterial species (which included E coli, Enterococcus spp, Pseudomonas aeruginosa, Acinetobacter baumannii, and methicillin-resistant S pseudintermedius) were susceptible to these drugs.21 In contrast, the isolated bacteria demonstrated susceptibility to other antibiotics (eg, chloramphenicol, imipenem, gentamicin, amikacin).21 These findings highlight the importance of submitting a sample from the infection site for culture and susceptibility testing for accurate identification of the causative bacteria and to guide appropriate antibiotic selection. A deep tissue sample (ideally without a collection of pus) is more likely to contain the true pathogens, rather than surface contaminants, and is preferred. Empiric (often 4-quadrant) therapy with a broad-spectrum antibiotic (eg, amoxicillin/clavulanate, ampicillin, enrofloxacin) may be initiated while waiting for culture and susceptibility results. Surgical intervention may be necessary if surgical site debridement is needed. In addition, a closed-suction drain may be placed to remove and prevent fluid accumulation.
Liposomal bupivacaine use for pain control has increased in veterinary medicine. A study evaluated surgical site infections after clean-contaminated and dirty GI surgeries with or without the use of liposomal bupivacaine; no association with higher rates of GI surgical site infections was found.20 Surgical technique, decreased surgical time, rigorous intraoperative hygiene protocols, and perioperative antimicrobial treatment can help reduce risk for surgical site infection (Figure 2).

FIGURE 2 Moderate peri-incisional erythema, edema, and bruising with serosanguineous discharge at the cranial and caudal end of the incision 4 days postoperatively in a dog after undergoing intestinal resection and anastomosis
4. Delayed Return to Eating
Monitoring patient appetite is a practical way of assessing postoperative progress and can be an important recovery marker after intestinal surgery. Early return to eating has been associated with improved outcomes; delayed nutrition can lead to prolonged hospitalization and slowed recovery. Human studies have found that initiating feeding within 24 hours after surgery, when clinically appropriate, is associated with enhanced recovery, reduced postoperative complications, decreased duration of hospitalization, improved nutritional status, and supported immune function.24 Immune system function is an important factor when healing from surgery. An estimated ≈60% to 70% of the immune system function in cats and dogs is in the intestines; an intact intestinal structure is therefore imperative for immune development.25 Changes in metabolism (eg, insulin resistance, decreased contractions within the stomach and small intestine) are evident after 24 hours of fasting.25,26
A structured feeding and monitoring plan in the postoperative period is essential to meet early caloric demands and can reduce mortality rates and shorten hospitalization.19 The resting energy requirement (70 × [body weight in kg]0.75) is an effective baseline for estimating daily caloric needs. A highly digestible GI diet is generally recommended as the initial diet of choice due to ease of absorption and low risk for GI irritation. In cases in which voluntary intake is inadequate, alternative nutritional support methods (eg, nasogastric, nasoesophageal, esophagostomy, or gastrostomy tube placement) should be considered to ensure consistent energy delivery. Close monitoring of appetite, GI motility, and feeding tolerance is critical to detect and prevent complications (eg, vomiting, regurgitation, aspiration).
Conclusion
Postoperative intestinal surgery complications (eg, intestinal dehiscence, ileus, surgical site infection, delayed return to eating) can significantly impact outcomes following intestinal surgery in dogs and cats. Early recognition, appropriate management, and prevention strategies are critical to improving prognosis. Meticulous surgical techniques, prompt nutritional support, and vigilant monitoring can reduce morbidity, support recovery, and enhance patient survival.