Epidural Injection & Catheter Placement
Overview
Several drugs can be administered into the epidural space of dogs and cats to provideanesthesia or analgesia in surgery, trauma, and critical care settings. The most frequently administered drugs are the local anesthetics and opioids,1-4 but α2-agonists (xylazine and medetomidine) and combinations of these drugs are also used.2,3
Advantages
Epidural injection requires lower drug doses than systemic injection, resulting in fewer side effects.
Less inhalant and/or injectable anesthetic is required in surgical patients, leading to improved cardiorespiratory function.
Use of lower doses and the long duration of action make epidural injection economical.
Indications
Anesthesia
Epidurally administered local anesthetics can be used as the sole agent for perianal, hind limb, and abdominal surgery. Injection of local anesthetics into the epidural space causes motor paralysis and sympathetic blockade. Sympathetic blockade may result in vasodilatation and hypotension, which can be severe in volume-depleted animals. In small animal practice, epidural anesthesia is rarely used as the sole method of anesthesia because animals are unlikely to tolerate physical restraint while fully awake. If this technique is used, the anesthetist must always be prepared to induce general anesthesia if the block is initially inadequate or wears off prematurely or if the animal stops tolerating the procedure. When used in conjunction with general anesthesia, epidural local anesthetics can significantly reduce anesthetic requirements and may prevent the "wind-up" phenomenon by blocking nociceptive input to the central nervous system (i.e., preemptive analgesia).
Analgesia
Epidural opioids provide long-lasting analgesia and cause fewer side effects than systemically administered opioids. They do not adversely affect motor function, so animals tend to retain function of the hind limbs. Epidural opioids alone are not sufficient for surgery, but frequently are used as a component of "balanced anesthesia." Given in enough volume, epidural morphine can provide analgesia as far forward as the thoracic limb5 and is a good choice for thoracic or fore limb surgery (e.g., thoracotomy, amputation, fibrosarcoma debulking, full chain mastectomies) in addition to hind limb or abdominal procedures. As with local anesthetics, epidural morphine reduces the requirements for inhalant agents and is beneficial for maintaining hemodynamic function during anesthesia.5,6 Epidural morphine, with or without long-lasting local anesthetics such as bupivacaine, can also be used to relieve pain associated with pancreatitis and peritonitis.3
Drugs
Local Anesthetics
One percent to 2% lidocaine or 0.25% to 0.75% bupivacaine can be used. Lidocaine lasts 60 to 120 minutes and bupivacaine 4 to 6 hours. Blockade up to L2 can be achieved with 1 ml per 5 to 10 kg of body weight in dogs; doses usually do not exceed 6 ml, even in very large dogs. Local anesthetics without epinephrine are recommended.
Opioids
Commercially available, preservative-free epidural morphine preparations contain 1 mg/ml. These drugs should be given at 0.1 to 0.2 mg/kg (Figure A). If repeated dosing is anticipated, preservative-free drugs are recommended because of the risk for preservative-related inflammation (neuritis). If a preservative-free formulation is not available, systemic (containing preservative) preparations can be used, but a new, sterile ampoule should be opened for each injection. The opioid dose can be diluted with local anesthetic, sterile saline, or sterile water to a final volume of 0.1 to 0.2 ml/kg (6 ml maximum total). Oxymorphone (0.1 mg/kg) has been used, but it is shorter acting than morphine and much of its effects result from systemic uptake. Buprenorphine has also been used epidurally in dogs (0.004 mg/kg) and is thought to provide analgesia similar to morphine.

Combination Protocols
Local anesthetics and opioids can be given concurrently. For example, morphine at 0.1 mg/kg can be combined with bupivacaine at 0.5 to 1.0 mg/kg. A complete listing of drugs and drug combinations for epidural use in dogs and cats can be found elsewhere.3
Contraindications
As with all medical procedures, there are some contraindications to epidural injection in dogs and cats. It should not be done in animals with bleeding disorders (disseminated intravascular coagulation, thrombocytopenia, von Willebrand's disease) because of the possibility of entering an epidural vessel with resultant bleeding and accumulation of large volumes of blood in the epidural space. Infection of the skin overlying the injection site is another contraindication because of the dangers of introducing infection into the epidural or subarachnoid space. Animals that are hypovolemic or hypotensive should not be given epidural local anesthetics until blood volume and blood pressure have been restored because of the vasodilatation that results from blockade of regional sympathetic nerves. Preexisting neurologic dysfunction and spinal trauma are additional reasons to consider alternative techniques for providing pain relief.
Although not an absolute contraindication, anatomical abnormalities (e.g., due to trauma or lumbosacral diskospondylitis) at the lumbosacral junction make this procedure much more difficult. Obesity also makes palpating landmarks challenging.
Complications
The incidence of reported complications associated with epidural injections is low, but problems can arise and may range from minor to serious and are well described.3 Most complications can be avoided by careful patient selection. Urine retention (7 out of 242 dogs)1 and pruritus (2 out of 242 dogs)1 have been reported after morphine administration, and there is a report of a dog developing an epidural abscess leading to diskospondylitis. Prolonged motor dysfunction (lasting longer than the expected duration of the injected local anesthetic) is occasionally reported, and it is possible that direct trauma caused by the epidural needle could damage the spinal cord and nerve roots. Conscious animals may struggle and resent injection of local anesthetics, and morphine can cause vomiting in conscious animals.
Although of no medical consequence, owners should be warned about delayed and abnormal hair regrowth at the clipped site over the lumbosacral space. The reasons for this phenomenon are not well understood.
Step-by-Step: How to Perform an Epidural Injection
What You Will Need
Clippers
Sterile prep solution
Sterile gloves
Appropriate drugs (see section on "Drugs" for doses)
Sterile saline or sterile water without preservative (bacteriostatic) agents (for test injection and "hanging drop" technique)
Glass (ideal but more difficult to obtain) or plastic syringe (to test for loss of resistance)
Injection Technique
1.5- to 3.5-inch, 20- to 22-gauge epidural or spinal needle with stylet*
3- to 6-ml syringe*
Catheter Technique (An epidural catheter should be considered if epidural analgesia will be required for more than 12 to 24 hours.)
Complete epidural catheter kit (Figure B) or
Tuohy or Hustead needle with catheter (Figure C)


* The size of the needle and syringe depend on the weight of the patient and drugs to be injected. A 1.5-inch, 22-gauge needle is suitable for cats and small dogs and a 2.5-inch, 20- to 22-gauge needle is recommended for larger dogs; a 3.5-inch, 20- to 22-gauge needle may be required for the largest breeds. A list of commercial sources and prices of supplies needed for epidural procedures can be found elsewhere.3
Anatomy and Landmarks
Step 1
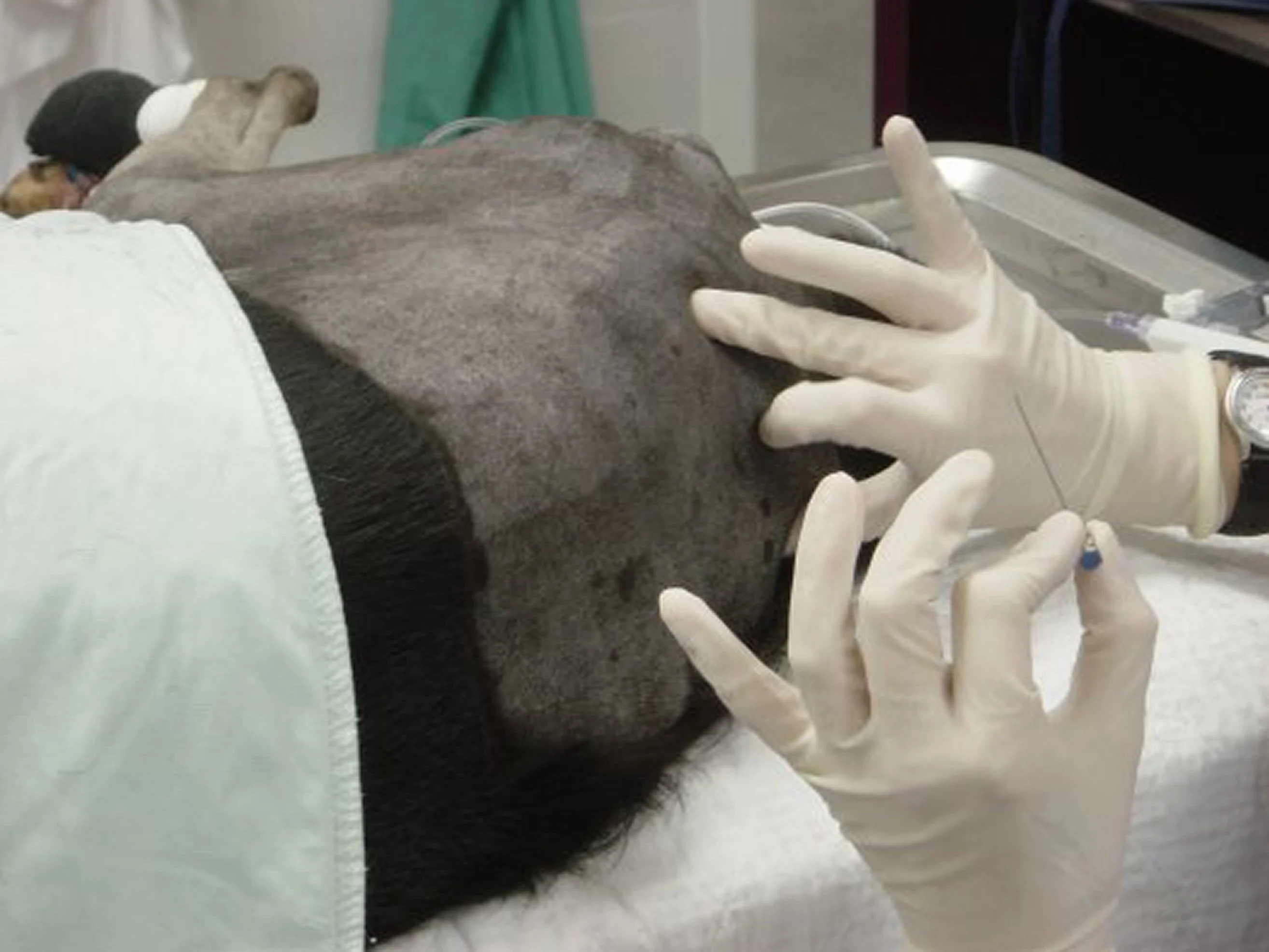
This procedure is most frequently done after induction of anesthesia but can be done on a sedated animal. Position the patient in sternal or lateral recumbency (Figures 1A and 1B). In either position, pull the hind limbs forward to "open" the space between the last lumbar vertebra and sacrum. The sternal position allows the "hanging drop" technique (discussed later) to be used to confirm entry into the epidural space, but it may not be suitable in animals with certain pathologic conditions of the pelvis or hind limbs, such as fractures or severe arthritis. In dogs and cats, the lumbosacral space (arrow) is the site for epidural injection (Figure 1C). In adult dogs, the spinal cord ends at approximately the 6th vertebra, but in puppies and cats, it extends more caudally, making a spinal/intrathecal/subarachnoid (CSF) injection more likely.



Dogs
Palpate the left and right dorsal iliac wings (arrows). Imagine a line between these two points; the injection site usually lies on or within 1 cm cranial or caudal of this line. Palpate the dorsal spinous processes. The 7th process (arrowhead) is shorter than the others (Figure 2A). The site for injection is just caudal to the 7th process, where a "dimple" may be felt, especially in small or thin animals.

Cats
The bony landmarks are generally easier to palpate in cats. However, the angle of the dorsal spinal processes is also steeper, and this should be kept in mind when palpating the landmarks (Figure 2B). The depth from the skin to the ligamentum flavum is less than in dogs, making it possible to place the needle too deeply.

Single Epidural Injection
Sterile Scrub
Perform a sterile scrub, put on gloves, and draw up your chosen drugs using a sterile technique. If the patient is awake, inject a small volume of fast-acting local anesthetic (i.e., lidocaine) intradermally and then subcutaneously. Using the landmarks outlined in Step 2, insert the spinal needle perpendicular to the skin, checking that it is on midline. Carefully advance the needle; a subtle "pop" may be felt as the tip penetrates the supraspinous, interspinous, and/or dorsal spinous ligaments. Continue to advance the needle until a second, more distinct "pop" occurs; this is the dense ligamentum flavum. The "pop" may vary with the patient, from barely perceptible to very noticeable. Once the ligament has been penetrated, the tip of the needle is in the epidural space. Remove the stylet and check for CSF or blood.
Procedure Pearl
A subtle "pop" may be felt as the needle penetrates ligaments. A second more distinct "pop" occurrs as it penetrates the ligamentum flavum.
Checking Placement
To check for correct placement, two techniques can be used. If the animal is in sternal recumbency, the "hanging drop" technique can be used. Once the stylet has been removed, fill the needle with sterile saline. Because the epidural space has a negative pressure, if the saline is "sucked" in, placement is correct (Figure 4). If the saline bubble is not drawn in, slowly move the needle while watching the bubble; when it disappears, check by adding more saline.
Procedure Pearl
If the needle tip is in the correct site, there will be no resistance to injection.

The second method is the "loss-of-resistance" technique. This technique can be used regardless of the animal's position. Lubricate a glass syringe with sterile saline, aspirate a few milliliters of air, and attach it (or a plastic syringe prepared in the same manner) to the spinal needle. If the needle tip is in the correct site, there will be no resistance to injection. Using the saline- and air-filled syringe, you should be able to inject easily with no change in the size of the air bubble, and the plunger of the syringe should not bounce back when released.
When correct placement is confirmed, inject the drugs at a slow, constant rate (this should take 60 seconds) to ensure even distribution of the drug.
Very few complications have been associated with epidural catheters placed for periods ranging from 1 to 7 days in dogs.7 There are no large-scale reports of their use in cats. Many different commercially available epidural catheter kits designed for humans, children, and animals have been used clinically in dogs and cats. The procedure is similar to that described above. However, the needle that comes with the kits is usually a Tuohy or a Hustead type, both of which are much duller than spinal or single-injection epidural needles and have a curved tip to control the direction of the catheter when it is being threaded through the needle.
Procedure Pearls
Tuohy and Hustead needles have a curved tip to control the direction of the catheter when it is being threaded through the needle.
An adaptor and a filter are applied to the end of the catheter to maintain sterility, and the catheter is secured by suturing to the skin.
Because of the bluntness of the needle, it may be useful to make a small skin incision (using local anesthetic if the dog is awake). Location of landmarks and confirmation of placement are as described in Step 4, paying close attention to sterility. The catheter is threaded through the needle (Figure 6A), and the tip can be placed where it will be most beneficial-for example, as high as the 4th thoracic vertebra for fore limb amputation or thoracic surgery. The catheters have radio-opaque, measured markings to assist in correct placement (Figure 6B). An adaptor and a filter are applied to the end of the catheter to maintain sterility, and the catheter is secured by suturing to the skin. An adhesive covering keeps the site clean and offers additional security for the catheter (Figure 6C).



Troubleshooting
Blood
If blood is encountered, the tip of the needle has entered an epidural venous sinus: Do not inject the drugs. Withdraw the needle and make a second attempt.
Cerebrospinal Fluid
If CSF is encountered, the tip of the needle has entered the subarachnoid space and there are two options:• Withdraw the needle and try again.• Perform a spinal/intrathecal/subarachnoid injection, but decrease the dose of drugs by at least 50% and monitor for more rapid onset and cranial distribution of the injected drugs. After spinal injection, be especially vigilant for respiratory depression.