Overview
Increased serum alkaline phosphatase (ALP) activity is a common finding in dogs and can be a result of primary hepatobiliary or bone disease, secondary to reactive hepatopathies, or caused by drug induction.1,2 Clinical presentation, clinical pathologic findings, and diagnostic imaging can be used as noninvasive aides in identification of the source of total ALP elevation.
Epidemiology
Incidence/Prevalence
In a study that included 1,022 blood samples from healthy and sick dogs, serum ALP activity increased in 39% of dogs (71% in dogs <1 year of age, 28% in dogs 1-8 years of age, 51% in dogs >8 years of age).3 The high incidence in dogs <1 year of age reflects increases in the bone isoenzyme because of osteoblastic activity in growing bone. The increase in dogs >8 years of age is likely associated with high incidence of benign nodular hyperplasia in older dogs.4
Signalment
Breed Predisposition5
Breed-specific hepatobiliary disorders suggest the possibility of a primary hepatobiliary disorder and should guide clinical decision-making.
Examples include
Shetland sheepdogs
Border terriers
Chronic hepatitis
Labrador retrievers
Doberman pinschers
Dalmatians
American and English cocker spaniels
English springer spaniels
West Highland white terriers
Bedlington terriers
Vacuolar hepatopathies
Scottish terriers
Miniature schnauzers
Age
Young dogs have increases in bone isoenzymes because of increased osteoblastic activity in growing bones.
Benign nodular hyperplasia is a common, age-related, incidental lesion in dogs; reported incidence ranges from 70% to 100% in dogs >10 years of age.4
Physiology
The primary clinical asset of serum total ALP determination is the ability (80%) to detect patients with hepatobiliary disease (ie, sensitivity).1,2,5,6 The major limitation in interpreting serum total ALP is low ability (51%) to exclude the presence of hepatobiliary disease (ie, specificity). The low specificity of serum total ALP is due to the presence of several ALP isoenzymes (ie, bone, liver, corticosteroid-induced) and the unique susceptibility of the enzyme to induction by drugs.1,2,5,6
Bone Isoenzymes
The bone isoenzyme of ALP accounts for approximately one-third of normal serum total ALP activity and is elevated in conditions associated with increased osteoblastic activity (eg, bone growth in young dogs) or in dogs with pathologic conditions (eg, osteomyelitis, osteosarcoma, renal secondary hyperparathyroidism).4 Serum bone ALP elevations in patients with these conditions are typically mild to moderate (3-5 times the upper limit of normal).1,7
Liver Isoenzymes
The liver isoenzyme of ALP is a membrane-bound enzyme present on biliary epithelial cells and hepatocytes. Increases in serum liver ALP activity are caused by elution of the enzyme from the membrane following hepatobiliary damage. The largest increases occur with focal or diffuse intrahepatic or extrahepatic cholestasis. Mild to moderate increases occur with chronic hepatitis and hepatic necrosis.
Corticosteroid-Induced Isoenzymes
The corticosteroid-induced isoenzyme of ALP is produced by the liver and found on hepatocyte membranes.1,2 This enzyme increases from de novo synthesis of the enzyme in dogs exposed to endogenous or exogenous corticosteroid excess.
Potential Causes of Changes in Serum ALP Activity
Determining the clinical significance of an increase in total serum ALP activity can be challenging. In dogs, ALP isoenzymes originate from the bone or the liver or can be induced by drugs in the absence of hepatobiliary damage (see Drugs Commonly Associated With Increased Serum ALP Activity). In addition, benign nodular hyperplasia, a clinically silent morphologic change in the liver, is a common cause of increased serum ALP in older dogs. An increase can also be caused by reactive hepatopathies that occur due to nonhepatobiliary disease in the splanchnic bed.1,2
Conditions in which total serum ALP activity may be elevated without the presence of clinically significant hepatobiliary disease are listed in Conditions Other Than Primary Liver Disease Associated With Increased Serum Total ALP Activity.
Drugs Commonly Associated With Increased Serum ALP Activity
Typical findings in dogs receiving corticosteroids include hepatomegaly, diffuse hyperechoic hepatic parenchyma on ultrasonography, vacuolar hepatopathy on hepatic biopsy, moderate to marked increases in total serum ALP and gamma-glutamyl transferase (GGT) activity with significant individual variability in the extent of induction (3-50 times the upper limit of normal) and little to no increase in serum transaminases (increase <2 times that of normal).12,13
Rarely, corticosteroids can be associated with hepatotoxicity.
Signs of possible hepatotoxicity include abnormalities of hepatic function tests (eg, increased total serum bile acids, hyperbilirubinemia, hypoalbuminemia, moderate to marked increases in serum transaminases).
Hepatic biopsy shows severe vacuolar hepatopathy with areas of focal necrosis, cholestasis, and ballooning degeneration of hepatocytes.12,13
Therapy involves treatment of hyperadrenocorticism or discontinuation of corticosteroids and substitution of an alternative immunosuppressive or anti-inflammatory agent.
Typical findings associated with enzyme induction caused by phenobarbital include increased total serum ALP activity, usually <5 times the upper limit of normal with normal to minimal increases in serum ALT, AST, and GGT activity.
Ultrasonography is typically normal, and most dogs do not have hepatomegaly.
Biopsy shows diffuse cytoplasmic granularity caused by proliferation of smooth endoplasmic reticulum.10,11
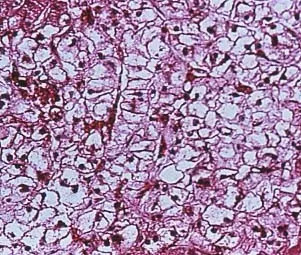
Phenobarbital is a known hepatotoxin. The liver of a dog with cirrhosis as a result of chronic hepatotoxicity from phenobarbital is shown in Figure 1.
Signs of hepatotoxicity include abnormal hepatic function tests (total serum bile acids, hypoalbuminemia, hyperbilirubinemia).
Elevations in serum ALT, AST, or GGT activity occur.
Hepatic biopsy shows chronic inflammatory/fibrotic disease.13
Treatment involves rapid tapering off from phenobarbital and changing to an alternative antiseizure medication, controlling the complications of hepatic failure, and initiating hepatoprotective therapy with ursodiol and/or S-adenosylmethionine.10,11

FIGURE 1 Drug-induced hepatopathy seen in the liver of a dog receiving phenobarbital antiseizure therapy
Multiple studies of dogs receiving cannabidiol preparations have shown that increases in serum ALP activity are possible, with a smaller number having serum ALT elevations.8,9
Evaluation to determine whether these increases in liver enzymes cause morphologic lesions has been limited.
Enzyme elevations are more common in dogs also receiving dual therapy with antiepileptic drugs or NSAIDs.8,9
Conditions Other Than Primary Liver Disease Associated With Increased Serum Total ALP Activity
Bone disorders
Young patients (normal physiologic-related finding)
Osteosarcoma
Osteomyelitis
Endocrinopathies
Diabetes mellitus
Hypothyroidism
GI disease
Pancreatitis
Inflammatory bowel disease
Hypoxia/hypotension
Congestive heart failure
Hypotensive crisis
Severe hemolytic anemia
Status epilepticus
Neoplasia
Hepatic metastasis
Paraneoplastic induction
Drug induction, particularly
Corticosteroids
Phenobarbital
Cannabidiols9,27,28
Systemic infections
Next Steps When Evaluating Patients With Abnormal Values
Patient History
History of drug administration
Particularly corticosteroids (oral, parenteral, or topical) or phenobarbital, but also other potentially hepatotoxic drugs (eg, cannabidiols, potentiated sulfonamides, NSAIDs)8-13
Both phenobarbital and corticosteroids can induce production of ALP but are also capable of causing hepatotoxicity. In cases of hepatotoxicity, serum transaminases not induced by these drugs are typically elevated.
Polyuria/polydipsia
Potential causes include hyperadrenocorticism, diabetes mellitus, chronic liver disease, and congenital portosystemic shunts.
History of dermatologic disorders
Potential causes include hyperadrenocorticism and hepatocutaneous syndrome.
Chronic intermittent GI signs
Potential causes include gastric ulceration secondary to chronic liver disease, congenital portosystemic shunts, chronic pancreatitis, and inflammatory bowel disease.
Physical Examination Findings & Potential Causes
Bone pain
Osteomyelitis, osteosarcoma
Diffuse cerebral signs
Hepatic encephalopathy from chronic liver disease or congenital portosystemic shunts
Potbelly
Abdominal wall muscle atrophy with centripetal redistribution of fat in hyperadrenocorticism
Jaundice
Prehepatic (eg, hemolytic anemia), hepatic, or posthepatic hyperbilirubinemia
Abdominal effusion
Chronic liver disease, neoplasia, pancreatitis, congestive heart failure
Hepatomegaly
Primary liver disease, vacuolar hepatopathy, passive congestion, hepatic lipidosis
Dyspnea/increased lung sounds
Congestive heart failure
Abdominal pain
Pancreatitis, cholecystitis, gastric ulceration
Relevant Diagnostic Testing, Findings, & Potential Causes14-22
Diagnostic Imaging
Radiography
Hepatomegaly
Vacuolar hepatopathy, congestive heart failure, hepatic lipidosis, focal and diffuse hepatobiliary disease
Microhepatica
Chronic end-stage hepatobiliary disease (eg, cirrhosis), congenital portosystemic shunts.
Choleliths
50% visible on radiographs, may be associated with secondary cholecystitis
Decreased abdominal detail
Ascites
Cardiomegaly and signs of pulmonary edema
Congestive heart failure
Lytic bone lesion
Bone tumor or infection
Normal liver
Does not rule out primary hepatic disease
Ultrasonography
Focal lesion
Hepatocellular carcinoma, hepatocellular adenoma, other cancers, abscess
Multifocal hepatic lesions
Benign nodular hyperplasia, vacuolar hepatopathy, metastatic disease, chronic hepatitis (eg, chronic granulomatous hepatitis)14,15
Diffuse hyperechoic liver
Vacuolar hepatopathy, hepatic lipidosis, lymphosarcoma
Diffuse hypoechoic liver
Passive congestion, lymphosarcoma, infectious hepatitis
Gallbladder/biliary tree
Gallbladder mucocele (diagnostic), distention of intra- and/or extrahepatic biliary tree, bile duct mineralization, choleliths17-19
Portal vasculature
Single or multiple acquired portosystemic shunts, portal vein thrombosis
Pancreas
Enlarged, hypoechoic, and surrounded by hyperechoic fat with pancreatitis
Thickened GI wall with retention of normal layering
Inflammatory bowel disease
Hepatic metastasis
Primary neoplasia of the spleen, stomach, pancreas, intestine, or adrenals
Honeycomb Liver
Hepatocutaneous syndrome20
Normal liver
Does not rule out primary hepatic disease
Laboratory Analysis
CBC
Serum chemistry profile
Urinalysis
Cytology of abdominal effusion
Hepatic biopsy
Identifies primary hepatic disease (eg, vacuolar disease [lipid or glycogen accumulation], neoplasia, vascular hypoperfusion, inflammatory/fibrotic disease)
Permits determination of hepatic copper levels to aid in diagnosis of copper-associated hepatopathy23
ALP isoenzyme analysis
Limited value
Determination of corticosteroid-induced increases in serum ALP via levamisole inhibition is a sensitive (95%) but not specific (18%) indicator of excess exposure to corticosteroids.
Many dogs with primary hepatobiliary disease have increases in both corticosteroid-induced isoenzymes and liver isoenzymes.
Corticosteroids induce increases in liver and bone isoenzymes, along with corticosteroid-induced isoenzymes.
Phenobarbital increases liver isoenzymes.24,25
Disease-specific testing
Hyperadrenocorticism
Low-dose dexamethasone suppression (LDDS) test
ACTH stimulation test
Hepatobiliary disease
Hepatic function test
Total serum bile acids
Blood ammonia levels
Prothrombin or partial thromboplastin time
Nodular hyperplasia
Hepatic function test
Hepatic biopsy
Disease-Specific Findings
Hyperadrenocorticism
Mild polycythemia, mild thrombocytosis, mild to moderate increase in serum ALP and GGT activity, hyperlipidemia, isosthenuria with mild proteinuria
Failure to suppress on an LDDS test, exaggerated response to an ACTH-stimulation test
Primary hepatobiliary disease
Concurrent increases in serum ALT, AST, and/or GGT; hyperbilirubinemia, hypoalbuminemia; low BUN; hypocholesterolemia; hypoglycemia
Abnormal hepatic function test
Elevated total serum bile acids
Hyperbilirubinemia with a normal packed-cell volume due to hepatic or posthepatic disease
Blood hyperammonemia
Confirms the presence of hepatic encephalopathy
Increased prothrombin time or partial thromboplastin time can accompany acute or chronic liver failure.14,15
Abdominal effusion consistent with a pure transudate or modified transudate on cytologic evaluation with chronic liver disease
Nodular hyperplasia
Subclinical patient, typically >8 years of age
Mild to moderate increase in total serum ALP activity that does not increase quickly over time
Other serum liver enzymes and hepatic function tests are normal.
Ultrasonography shows multifocal nodules.
Hepatic biopsy shows well-circumscribed nodules with normal but often vacuolated hepatocytes surrounded by normal hepatic tissue.
Wedge biopsy is optimal because nodular hyperplasia must be differentiated from a regenerative nodule in a cirrhotic liver, which requires fibrosis and/or inflammatory changes in the surrounding hepatic parenchyma.4
Acute pancreatitis
Abdominal effusion consistent with acute, nonseptic neutrophilic inflammation
Increase in serum amylase and/or lipase or immunoreactive pancreatic lipase
Diabetes mellitus
Persistent hyperglycemia
Malignancy
Malignant effusion, high protein fluid with exfoliated neoplastic cells (pancreatic, intestinal, adrenal adenocarcinoma, or lymphoma), absence of neoplastic cells (does not rule out cancer), hemorrhagic effusion with ruptured hemangiosarcoma
Diagnosis at a Glance
Measurement of total serum ALP activity is a sensitive but relatively nonspecific indicator of hepatobiliary disease. Specificity for primary hepatobiliary disease is increased when used in series with other serum liver enzymes.
Hepatobiliary, bone, and corticosteroid-induced isoenzymes contribute to total serum ALP levels in dogs, but isoenzyme analysis is rarely clinically useful.
Elevation in the bone isoenzyme of ALP is associated with increased osteoblastic activity. In patients with osteosarcoma, the degree of serum bone ALP elevation predicts survival time.
Definitive diagnosis of induction of serum ALP is made via medication history, clinical or laboratory signs suggestive of hyperadrenocorticism, and abnormal LDDS or ACTH stimulation test results.
Benign nodular hyperplasia is the most common reason for mild to moderate, very slow progressive increases in serum ALP activity in older dogs.
Abdominal ultrasonography is particularly useful in helping determine the cause of increases in serum ALP activity in dogs.
In some cases, ultrasonography can be diagnostic (eg, for gallbladder mucoceles, hepatic tumors, pancreatitis, extrahepatic bile duct obstruction) or can raise the index of suspicion for specific conditions (eg, benign nodular hyperplasia, cholecystitis, cirrhosis).
Advanced CT or MRI imaging may help improve diagnostic accuracy, particularly for evaluation of mass lesions.22,26
Treatment
Treatment should be based on the specific cause of the increase in serum ALP activity.
Monitoring & Follow-Up
Subclinical Patients
Observation for signs of occult hyperadrenocorticism; LDDS or ACTH-stimulation test should be performed.
If there are no signs of another disease process and total ALP is <5 times the upper limit of normal, total serum ALP activity should be monitored periodically.
The typical pattern of benign nodular hyperplasia is a very slow progression in serum ALP activity over many years without increases in other serum liver enzyme activity.
If total ALP is persistently elevated, abdominal ultrasonography should be performed and evaluated.
Multiple nodules with normal to increased liver size are most consistent with benign nodular hyperplasia but can also be due to metastatic disease or round cell neoplasia. Failure to identify a primary tumor on abdominal ultrasonography helps differentiate benign nodular hyperplasia from metastatic disease. Dogs with round cell disease are not typically subclinical.
Multiple nodules in a small liver with irregular margins is consistent with a diagnosis of cirrhosis. Laparoscopic hepatic biopsy is needed to confirm the diagnosis.
Focal mass
Percutaneous biopsy or fine-needle aspiration
Gallbladder mucocele
Evaluation for medical versus surgical (cholecystectomy) management17,18
Choleliths (without obstruction) with evidence of gallbladder wall pathology
Cholecystocentesis with bacterial culture and sensitivity of bile or therapeutic trial of antibiotics and choleretics (ursodiol)
Patients With Clinical Signs
Diagnostics for detection of a primary or secondary hepatobiliary disorder (eg, hepatic tumor or abscess, gallbladder mucocele, cholecystitis, chronic hepatitis, cirrhosis, pancreatitis, extrahepatic bile duct obstruction), typically including CBC, serum chemistry profile, urinalysis, and abdominal ultrasonography, should be performed.
Additional hepatic function tests (eg, total serum bile acids, blood ammonia) may be necessary.
Advanced imaging or hepatic biopsy may be necessary for definitive diagnosis.23,26
Prognostic Significance
Increases in serum ALP activity in dogs with primary hepatobiliary disease are indicative of active hepatobiliary disease. Increase is usually, but not always, proportional to the severity of ongoing damage. In patients with end-stage fibrotic liver disease, serum ALP activity may not be elevated in proportion to the degree of hepatic disease because of enzyme depletion secondary to replacement of normal hepatocytes by fibrosis.
The liver has a large regenerative capacity and great functional reserve, thus the magnitude of elevation of serum ALP is not indicative of the degree of functional impairment and is not prognostic; however, the prognostic significance of total serum ALP can be improved by sequential evaluation, especially in conjunction with hepatic biopsy or function tests. The half-life of serum ALP in dogs is 72 hours; a 50% decrease in total ALP over a 3- to 4-day period may therefore indicate resolution of acute injury. In the absence of hepatotoxicity, serum ALP elevations caused by phenobarbital should return to normal 2 to 4 weeks after discontinuation of the drug. Increases in total ALP due to corticosteroid excess, however, may take several months to normalize.
Elevations in bone ALP are associated with shorter survival times in patients with appendicular osteosarcoma.7