Top 5 Dermatoses Associated with Electromagnetic Radiation
Alexander Werner Resnick, VMD, DACVD, Animal Dermatology Center
Electromagnetic radiation (EMR) is caused by the motion of electric charges in waves that produce a magnetic field. EMR encompasses the range of frequencies (ie, wavelengths) in the electromagnetic spectrum that extend from below radio waves to the gamma radiation used for cancer therapy and beyond. Depending on the insult, energy transferred by radiation can affect the skin by direct damage, exposure to extreme temperature, or both.
Top 5 Dermatoses Associated with Electromagnetic Radiation
1. Solar Dermatitis
2. Acute Thermal Injury
3. Chronic Thermal Injury
4. Frostbite
5. Radiation Therapy
The varied hair coat and pigmentation of dogs and cats can both protect them from EMR and allow damage to the skin from much of the solar spectrum. The solar spectrum consists of 3 types of light (from long wavelength/low energy to short wavelength/high energy):
Infrared light (700 nm-1 mm)
Visible light (400-700 nm)
Ultraviolet (UV) light (100-400 nm)
In comparison, gamma radiation used to treat neoplasia consists of high-energy photons and is <10 picometers in wavelength.
Following are the top 5 cutaneous insults caused by excessive (or lack of) EMR.
1. Solar Dermatitis
Solar dermatitis (ie, sunburn) is caused by direct UV light injury to skin cells. Both UVA, which is least absorbed by the ozone layer and falls just above the range of visible light at 320-400 nm, and UVB (290-320 nm) cause skin damage; UVC (100-290 nm) is completely absorbed by the ozone layer and does not reach the skin.1
Related Article: Top 5 Sun-Induced Skin Lesions in Dogs
The severity of solar dermatitis is related to the form, duration, and intensity of sun exposure. UVA penetrates the epidermis and causes the greatest damage at the basal keratinocyte layer and the dermis; this is the main cause of photoaging and wrinkling. UVB primarily penetrates the epidermis. This can produce reddening and inflammation and can promote development of cutaneous cancers.1 Acute exposure to UV light can damage keratinocytes, alter the skins immune system, and cause keratinocyte proliferation and mutation.2
Solar dermatitis frequently occurs on the dorsal muzzle (nasal) in dogs and cats, as well as in other nonpigmented regions (eg, eyelid margins, lip margins) and on the ventrum (truncal) in dogs. Nasal lesions begin as erythema and scaling at the junction of the nasal planum and dorsal muzzle and expand caudally as inflammation causes adjacent hair loss, which exposes the skin to additional UV light. Subsequent scarring and postinflammatory depigmentation can further dermatitis development (Figure 1), and lesions can wax and wane depending on seasonal sunlight fluctuations.

Chronic solar dermatitis in a 7-year-old male dalmatian. The pigmented nasal planum and periocular skin are not affected. The nonpigmented skin of the dorsal muzzle is scarred and excoriated.
Australian shepherd dogs are most at risk for nasal solar dermatitis.2,3 The primary differential for canine nasal solar dermatitis is discoid lupus erythematosus, which can cause depigmentation of the nasal planum followed by crusting.
Canine truncal solar dermatitis occurs in breeds and individuals with light pigmentation and an inclination toward sunbathing, especially dalmatians, bull terriers, boxers, bulldogs, basset hounds, beagles, and American Staffordshire terriers.2 The exposed side, depending on a dogs tendency toward right or left lateral recumbency, is more affected than the down (ie, sheltered) side.

Lateral aspect of a 3-year-old boxer with truncal solar dermatitis. The islands of pigmented skin are less affected, as demonstrated by hair growth. These areas were palpably less scarred than surrounding nonpigmented skin. Lichenification can be noted in the erythematous areas.
Initial lesions of truncal solar dermatitis consist of mild erythema with subtle lichenification and scaling of the axillae and flank regions (Figure 2). After repeated exposure and thickening of the skin, comedones can develop with accentuation of the follicular ostia. This can cause deep pyoderma and scarring. Dogs with chronic UV light-damaged skin are at increased risk for developing sun-induced hemangioma, hemangiosarcoma, and/or squamous cell carcinoma4; white or light-colored cats are also predisposed to squamous cell carcinoma, typically on the eyelid margins or lip margins.
Solar dermatitis is treated primarily by avoiding sunlight; sunblock should be applied to prevent further damage. Secondary infection, if present, should be treated with appropriate antibiotics. UV light can cause keratinocyte apoptosis and cutaneous inflammation induced by prostaglandins that also promote tumor growth.5 Cyclooxygenase (COX) converts arachidonic acid into prostaglandin, and COX-2 is specifically overexpressed in actinic keratosis and squamous cell carcinoma.6 In a recent study, the COX-2 inhibitor firocoxib (5 mg/kg once a day) was demonstrated to promote the regularization of epithelial proliferation and was associated with improvement of clinical signs in dogs with solar dermatitis.7
2. Acute Thermal Injury

Acute thermal burn from a heating pad during surgery on the dorsum of an adult pitbull crossbreed. The central zone of necrotic tissue is separating from the subcutis. The injury occurred 7 days before presentation.

Acute solar thermal burn 2 weeks postexposure in a 5-year-old border collie crossbreed. Sun exposure from walking outside on a hot day caused injury to the skin beneath the black hair coat.
Burns may be superficial, partial-thickness, or full-thickness. The extent and depth of burns are determined by the intensity and length of exposure to heat. Thermal burns are characterized by a central zone of tissue coagulation surrounded by an area of ischemic necrosis caused by circulatory stasis and bordered by an area of erythema. A common cause of acute thermal burn injury in dogs and cats is electric heating pads (Figure 3).2Darkly pigmented dogs can develop acute thermal burns from prolonged sun exposure exacerbated by high ambient temperature (Figure 4). Burns may also occur from exposure to microwave radiation and electricity.8
Related Article: Basic Wound Care
On glabrous skin, burns are apparent immediately; lesions hidden by a hair coat may go unnoticed for >72 hours. The central necrotic zone ultimately sloughs to reveal a deep defect. Treatment involves repeated tissue debridement and prevention of secondary infection. Superficial and partial-thickness burns may heal completely through re-epithelialization from hair follicles and sebaceous glands. Full-thickness burns heal with extensive scarring because of the destruction of all epithelial structures.
3. Chronic Thermal Injury

Erythema ab igne on the ventrum of a 16-year-old domestic shorthaired cat caused by sleeping on a heated mat. Affected areas are alopecic and thickened. They appear red-brown in color but are not inflamed.
Skin can become damaged by prolonged exposure to low levels of infrared radiation from sources such as heat registers, electric blankets or mats, tube-type televisions, and laptop computers. The pathogenesis of chronic thermal injury, also known as erythema ab igne, is unclear but likely involves damage to superficial blood vessels leading to vascular dilation, extravasation of red blood cells, and deposition of hemosiderin. This can cause the appearance of hyperpigmentation.9
Lesions occur in areas of contact with radiant heat, most often on the ventrum, and consist of occasionally erythematous, irregularly shaped but well-demarcated patches of alopecia with scarring, variable hyperpigmentation, and mild scaling (Figure 5). Chronic thermal injury is benign, and removing contact with the heat source should be curative.
4. Frostbite
Although not caused by EMR, frostbite results from the absence of sufficient thermal radiation and thus is included on this list. Exposure to cold can cause peripheral vasoconstriction, which can limit blood flow to the skin. Prolonged exposure to cold below 32F (0C)or contact with a frozen objectcan cause direct cell damage, damage from ice crystals that form in and around tissues, and/or damage from ischemia caused by vascular constriction and thrombi.

Loss of the pineal tip caused by frostbite in an adult domestic shorthaired cat. Both pinnae were affected. Photo courtesy of Karen Moriello, DVM, DACVD.
In dogs and cats, the tail tip, pinnae, digits, and scrotum are most at risk for frostbite (Figure 6). Frozen skin appears pale; necrosis followed by sloughing occurs as the skin thaws. Treatment is similar to that for thermal burns, with debridement as well as protection from cold to prevent further injury.
Related Article: Hypothermia Overview
5. Radiation Therapy
Radiation therapy uses ionizing, high-energy waves (eg, x-rays, gamma rays) to destroy cancer cells. Ionizing radiation directly damages DNA, which can cause cell death and other effects. To provide a large dose of radiation to the tumor while reducing the exposure of surrounding tissues, radiation beams are shapedor aimedfrom several angles to intersect at the tumor. Fractionation of dosing also allows normal cells to recover between treatments; cancer cells are slower to recover. Because of its rapid turnover rate, however, the epidermis is especially sensitive to acute radiation-induced dermatitis, with lesions developing in >90% of dogs receiving fractionated radiation therapy for skin and soft-tissue tumors.10

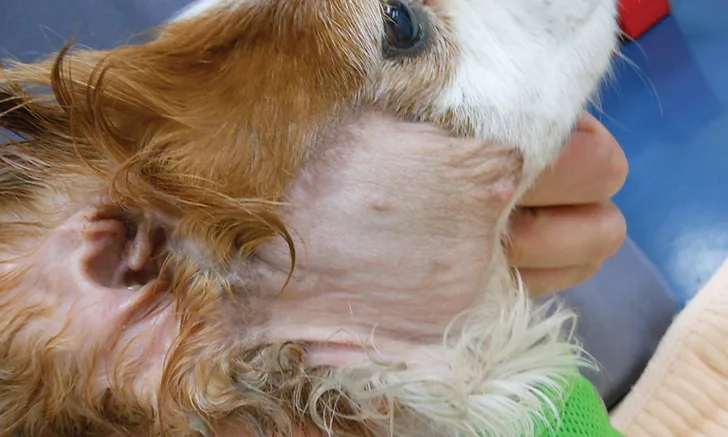
Scarring from radiation therapy for squamous cell carcinoma of the external ear canal in a 9-year-old Cavalier King Charles spaniel. The distinct borders indicate the treatment area.
Lesions can develop several weeks into therapy. The skin becomes pink and tender before mild exudation and eventual alopecia with scarring (Figure 7) appear. Striking leukotrichia is common. Secondary bacterial infection may occur in the immediate posttreatment period. Scarring often persists after discontinuation of radiation treatments.
Related Article: Advancing Oncology with Radiotherapy
Conclusion
The skin presents a barrier to the outside environment. It must be tough yet flexible and solid yet permeable. EMR can move through the hair coat, past the pigmentation, and into the cells to cause mutation and destruction. Protection from excessive exposure to heat and cold, UV light, and gamma rays is the best way to protect the skin against these important dermatoses.
COX = cyclooxygenase, EMR = electromagnetic radiation, UV = ultraviolet