Top 5 Considerations for Anesthesia of a Geriatric Patient
Patrick M. Burns, BVSc, MANZCVS, DACVAA, University of Montreal
It is difficult to define exactly what constitutes a geriatric patient. The use of a particular age does not take into account the different life expectancies of the various breeds and subsequent changes in physiological reserve as the patient ages. Some have suggested a definition based on a specific fraction (75%-80%) of the breed’s life expectancy.1 This definition assumes that the linear degradation of the physiological reserve is the same for all individuals.2-4 Changes in physiological reserve—not patient age—affect anesthesia.
1. Pre-anesthetic Evaluation
A pre-anesthetic evaluation forms the basis of an anesthetic protocol, and as such, its importance cannot be overstated. A thorough history and a physical examination are key to formulating an anesthetic protocol (see Common Comorbidities). Evaluating the oral intake and metabolic outputs of a patient and the maintenance of a normal body weight is a general indicator of overall health; however, this tends to evaluate the metabolic function of the patient only. Changes in behavior or exercise tolerance can point to more serious problems with the CNS or cardiovascular system. CBC, serum chemistry panel, and urinalysis can help reinforce clinical suspicion of any problems detected on physical examination as well as identify some of the age-related diseases and sub-clinical degenerative organ dysfunction more likely to occur with aging. Differentiating geriatric patients with benign, normal age-related changes from those with degenerative organ dysfunction or clinical disease allows appropriate modification of anesthetic protocol.2
Common Comorbidities
Obesity31,32
Osteoarthritis
Hypertension33-35
Valvular and myocardial diseases11,33,36
Renal disease36
2. Modify the Anesthetic Protocol
In geriatric patients, there are two major factors that influence anesthetic protocol. The first is degenerative organ dysfunction (Table 1). In general, degenerative organ dysfunction—particularly in the brain, liver, and kidneys—may cause more profound pharmacodynamic effects to anesthetics, delay in the onset of action, prolongation of the duration of effect,5 and predispose the patient to an increased risk for adverse effects. Consequences of this can be seen in evaluation of perioperative small animal fatalities where the risk for mortality for small animals over 12-years of age is increased by a factor of 7.6 One study investigating the pharmacokinetics of propofol observed that geriatric dogs were more sensitive to the effects of propofol and therefore required a lower dose.7 Ideally, a drug’s pharmacokinetic profile should not be influenced by the metabolic status of the patient. This is one reason why propofol and alfaxalone have more predictable pharmacodynamic effects than thiopental in patients with metabolic derangements.8
The second factor influencing protocol involves age-related diseases. It is important to identify the main physiologic- and metabolic-induced perturbations and choose a protocol that will minimize impact on compensatory mechanisms (Table 1).
As a general rule, if any doubts exist as to the physiologic or metabolic capacity of the patient, use premedicants, which have minimal effects on blood flow and can be antagonized short-acting drugs (eg, midazolam, fentanyl), followed by short-acting drugs (eg, propofol, alfaxalone, isoflurane).
Table 1: Specific Systems & Physiologic Changes That Can Affect Anesthetic Protocol
3. Age-Related Physiologic Changes
Neurologic System
In general, geriatric patients may respond more profoundly to sedatives and anesthetic agents.7 This is thought to be because of reductions in cerebral blood flow, decreases in neurotransmitters, and receptor affinity as well as changes in drug pharmacodynamics.3,5,9-10 It may be prudent to reduce the dosage and to allow for a longer duration of action.2
Cardiovascular System
Co-existence of primary cardiac disease3,9,11 and cellular structural changes9,12 are prevalent in geriatric patients; however, in some species (eg, Cavalier King Charles spaniels,13,14 bull terriers15), this prevalence is greater than 50%. In general, these physiologic changes render the cardiac reserve intolerant to changes in cardiovascular function. Both heart rate and blood pressure should be kept within normal range. Use of benzodiazepines, opioids, and locoregional techniques can all minimize the deleterious effects induced by anesthetic agents.3,9,10,12,16
Respiratory System
In one study, no differences in arterial blood gases were identified when comparing unanesthetized dogs over 8 years of age to younger dogs17; however, changes to pulmonary mechanics, notably thoracic compliance,16 and ventilatory responses to hypercapnia and hypoxemia9 have been observed. Anesthesia and these mechanical changes to pulmonary mechanics reduce the capacity of geriatric patients to compensate for changes in oxygenation and ventilation. Mechanical ventilation is frequently required to maintain the PaO2 and PaCO2 within normal limits during anesthesia, especially in obese patients.10,12,16
Renal System
Reductions in renal blood flow, glomerular filtration rate,10 and disturbances to sodium and water homeostasis9 render geriatric patients intolerant to hypovolemia and fluid overload.10 Using fluid therapy to maintain a normal urine output is questionable.18,19 General anesthesia causes a reduction in urine output. Trying to maintain a normal urine output during anesthesia may also result in edema, hemodilution, hypoproteinemia, electrolyte disorders, and (potentially) coagulopathies.18 Fluid therapy can cause deleterious effects if used injudiciously.20,21 A prudent approach to hypotension is to give a fluid bolus of 10-20 mL/kg over 15 minutes and optimize the depth of anesthesia, then reassess the blood pressure, hematocrit, and total protein. If the blood pressure is still low, then a perfusion of a positive inotrope such as dobutamine may be instituted.3,9,16 This approach is assuming the original blood volume was normal.
Hepatic System
Liver mass as well as synthetic and metabolic capacities reduce with age.9,10 Body composition also changes with age. Muscle mass decreases and fat content increases. Both of these changes alter the pharmacokinetics and pharmacodynamics of medications.2 Decreases in cardiac output can also prolong duration of action of various medications metabolized by the liver.3,9,10 It may be necessary to reduce initial dose of drugs and to alter dosing intervals.19,20,22
4. Minimizing the Depth of Anesthesia
A multimodal approach to general anesthesia can minimize the depth of anesthesia, which reduces cardiovascular depression induced by anesthetic agents. This improves arterial blood pressure and oxygen delivery. Degenerative changes to the baroreceptor reflex and cardiac function of geriatric patients increase susceptibility to hypotension during anesthesia.1,9,10,23,24 Incorporating a multimodal analgesic plan such as one including nonsteroidal antiinflammatory drugs, opioids, and/or locoregional anesthesia into the anesthetic protocol can minimize inhalant anesthetic requirements. The most effective approach is to include locoregional anesthetic techniques such as a dental block (Figure 1). These have the added benefit of blocking the stress response induced by the surgical intervention.
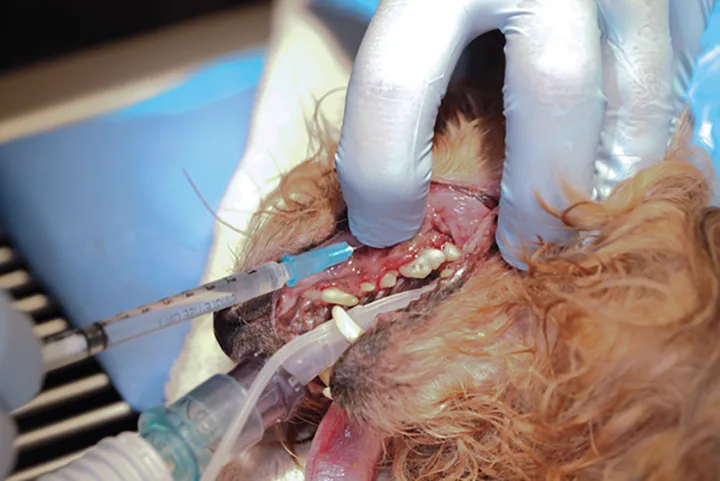
An infra-orbital nerve block is a good example of how locoregional anesthesia can inhibit nociception associated with the dental procedure. This permits reduction of the depth of inhalant anesthesia and improves cardiac output and wound healing if administered during the post-operative phase.
5. Continue Monitoring During the Recovery
It is important to continue monitoring a patient until it can protect its own airway and is normothermic. Hypothermia, hypotension, and hypercapnia can prolong the recovery phase and can exacerbate the potential for arrhythmias.1,9,10 Respiratory support during this time can accelerate the rate of elimination of inhalant anesthetics (Figure 2). Delayed metabolism of drugs prolongs the recovery phase. A benzodiazepine or opioid antagonist (eg, flumazenil or naloxone, respectively) may be required if profound sedation is suspected and metabolic disturbances have been ruled out. Both classes of drug can cause emergence delirium. This is especially true of patients experiencing little pain or discomfort after surgery. They are more likely to experience emergence delirium caused by the residual effects of the sedatives and analgesics. Emergence delirium can also be exacerbated by cognitive dysfunction, which has been reported in humans, cats, and dogs.9,25-27 Pathological changes have been observed similar to human Alzheimer disease28,29 and age-related storage diseases of the brain.23,24 Even changes in environment can cause significant uncharacteristic behaviors and poor recoveries.9,10
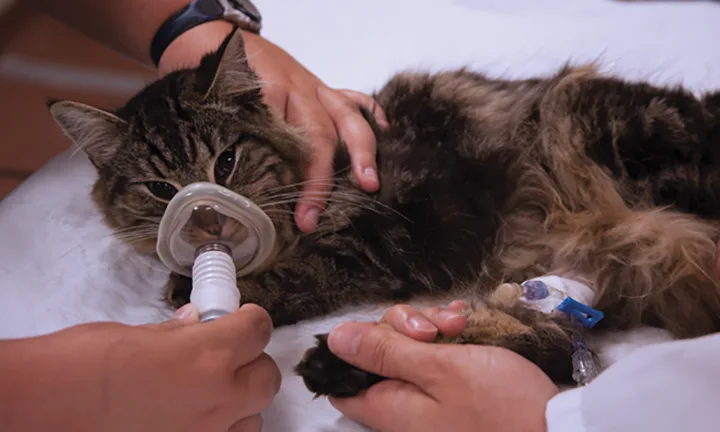
Oxygen therapy during all phases of anesthesia can reduce the 2 risk for arrhythmias.
If drug-induced dysphoria is suspected, it is not necessary to fully antagonize their effects but to partially antagonize effects until the patient returns to a state where it is comfortable and can function normally. Based on the experience of the author and others, both flumazenil and naloxone30 have been used to treat emergence delirium. Used at 0.001-0.005 mg/kg IV to antagonist their respective agonists, they are given in small intravenous boluses every 3 minutes until the patient is calm. When antagonizing opioids, it is necessary to monitor for signs of pain (Table 2).
Table 2: Pain vs Dysphoria or Emergence Delirium*
*Final diagnosis is based on events leading to the recovery (eg, response to analgesics during surgery) and the various medications given during this time. History, physical examination, and response to therapy must be considered. This may be highly subjective and extremely difficult in patients with co-existing neurological diseases. If doubt exists, it is better to assume that the patient is in pain and administer a short-acting opioid (eg, fentanyl, remifentanil) and evaluate the response. The signs and observations here are suggestive only and must be put in context with each individual situation.
**Opioid administration in cats can cause mydriasis; this could be a sign of opioid-induced dysphoria in the cat.
†Only administer the antagonist if the agonist was previously administered.
Closing Thoughts
The pre-anesthetic evaluation is important in identifying potential age-related diseases and organ dysfunction. An anesthetic protocol with minimal cardiovascular effects helps ensure hepatic and renal perfusion, which optimizes drug metabolism. Age-related physiologic changes and their potential impact on analgesic and anesthetic agents must be appreciated. Maintaining oxygen delivery is key to successful management of the geriatric patient. A multimodal analgesic plan can reduce the anesthetic requirements and thus reduce adverse effects of inhalant anesthesia such as vasodilation, reduced contractility of volatile anesthetics, and elevated or reduced heart rate. Geriatric patients may be more likely to develop a problem during anesthesia; this makes monitoring all the more important.