Methicillin-Resistant Staphylococcal Infections
Staphylococcus species are part of the normal mammalian and avian flora, but the worldwide increase in methicillin-resistant infections is likely a reflection of increased antibiotic use.
The Big Misnomer. Although methicillin resistance is the common term, today oxacillin is the antibiotic used for susceptibility testing, as methicillin is no longer manufactured and oxacillin is considered to be more stable.
Profile
Definition
Staphylococcus bacteria (gram-positive, facultative, aerobic cocci) are opportunistic pathogens that can cause skin, body cavity, and soft tissue infections when inflammation is present and/or the host has compromised defense mechanisms.
S aureus, S schleiferi, and isolates of the S intermedius group (SIG; ie, S pseudintermedius, S intermedius, S delphini) are important organisms in veterinary medicine.
Molecular testing has determined that the primary pathogen of dogs is S pseudintermedius; previous S intermedius isolates are now believed to have been S pseudintermedius.
The occurrence of methicillin-resistant staphylococcal infections is increasing.
Methicillin resistance is encoded by the mecA gene, which confers intrinsic resistance to all b-lactam antibiotics (ie, penicillins, cephalosporins).
The gene is located on the staphylococcal chromosomal cassette along with other resistance genes, allowing resistance to multiple antimicrobials.
In humans, methicillin-resistant S aureus (MRSA) can be hospital- or community-acquired.
Human or animal S aureus carriers without clinical signs are classified as colonized.
Studies have shown that up to 30% of humans have nasal colonization of S aureus; approximately 0.5% of those isolates are MRSA.1,2 The percentage of isolates of the methicillin-resistant S intermedius group (MRSIG) is low (<0%–5%), depending on the study and where it was conducted.1
Systems
Skin, soft tissue, and the ears are the most common sites of methicillin-resistant infection; however, any organ has the potential for infection.
Geographic Distribution
Methicillin resistance has been noted worldwide in humans and animals, but prevalence of MRSA and MRSIG varies widely with geographic distribution.
Methicillin-resistant S pseudintermedius (MRSP) can be found in or on clinically healthy dogs and cats. Reported prevalence of colonization ranged from 1.5% to 17% in healthy dogs and in healthy cats was 1.2%.3
Signalment
Methicillin resistance can occur in dogs and cats of any age, breed, or sex.
Transmission & Risk Factors
Pets are most likely to contract S aureus or MRSA from humans.
Studies have shown that less than 4% of healthy dogs and cats are colonized with MRSA.
Risk factors for MRSA in pets include infections related to surgery, hospitalization, prior antibiotic use, or exposure to humans with MRSA. Therapy pets in hospital visitation programs are also at increased risk.
Pets typically acquire MRSP via animal-to-animal contact; healthy colonized pets are the most likely source.
Signs
Clinical signs of methicillin-resistant infections are identical to methicillin-sensitive infections. Clinical suspicion of methicillin resistance includes lack of response to appropriate empiric therapy, worsening of signs while receiving therapy, and/or recurrent infections (particularly of the skin and ears).
MRSA = methicillin-resistant S aureus, MRSIG = methicillin-resistant S intermedius group, MRSP = methicillin-resistant S pseudintermedius, SIG = S intermedius group
Diagnosis
Definitive
Speciation of S pseudintermedius, S intermedius, and S delphini requires molecular testing; most laboratories will report SIG isolates.
S schleiferi and S aureus can be identified by standard laboratory speciation test kits (eg, API Staph, biomerieux-diagnostics.com).
Methicillin resistance requires culture and susceptibility testing via broth or agar-based tests.4
Accurate detection of methicillin resistance can be difficult because of heteroresistance (ie, the presence of 2 subpopulations [one susceptible, the other resistant] that can coexist within a culture).
These cells grow more slowly than oxacillin-susceptible populations and longer incubation periods are required to avoid missing the diagnosis.5
PCR assay is considered the gold standard for detection of the mecA gene.
Other Diagnostics
Many methicillin-resistant staphylococcal infections in dogs and cats are complicated by secondary yeast overgrowth; cytologic examination of glass microscope slides or clear acetate tape is useful to identify organisms.
MRSIG Illustrated
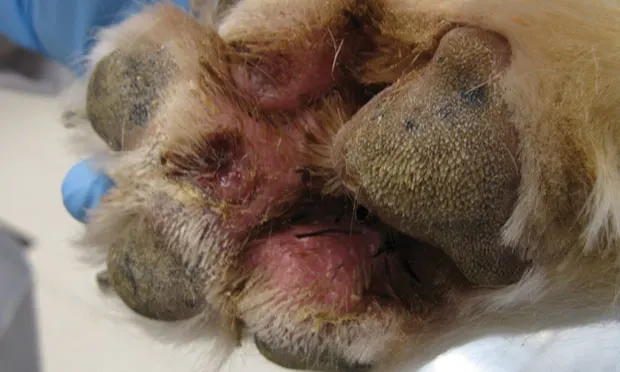
A dog was presented with a 3-month history of severe lameness and swelling of the lower leg. Culture of a draining tract between the foot pads (A) revealed a MRSIG infection susceptible to chloramphenicol. After 5 days of treatment, the paw is markedly improved (B). The dog made a full recovery. (View larger image)

MIC = minimum inhibitory concentration, MRSA = methicillin-resistant S aureus, MRSIG = methicillin-resistant S intermedius group, MRSP = methicillin-resistant S pseudintermedius, MRSS = methicillin-resistant S schleiferi, PCR = polymerase chain reaction, SIG = S intermedius group, VAST = Veterinary Antimicrobial Susceptibility Testing [subcommittee]
Treatment
Inpatient/Outpatient
Inpatient care is indicated only if the animal requires surgery, surgical debridement, or supportive care because of organ involvement.
Medical
All MRSIG/MRSA infections are resistant to b-lactam antibiotics; some strains may be resistant to other antibiotics.
Systemic antibiotic selection must be based on culture and susceptibility testing. Length of treatment will depend on severity of infection but is usually no less than 21 to 30 days.
Topical and local therapy (antibacterial shampoos, ointments, incision, flushing of abscesses) may be beneficial.
Secondary yeast infections, as well as primary or secondary seborrhea, should be treated appropriately.
Grooming and clipping hair from affected areas are crucial.
Decolonization of healthy animals with MRSA is not necessary and is not recommended for healthy animals with MRSP or methicillin-resistant S schleiferi (MRSS).
Careful search for the underlying trigger is critical, particularly when the patient history includes use of multiple antibiotics and/or chronic skin or ear disease.
Activity
Limiting activity is not indicated unless the infected area requires immobilization or bandaging.
nfected or colonized dogs should be exercised where they will not encounter other dogs.
Infected or colonized cats should be kept indoors.
Medications
Topical Therapy
Therapy with benzoyl peroxide shampoo or q24h or q48h chlorhexidine shampoo is helpful as adjuvant therapy in pets receiving systemic antibiotics.
Multidrug-resistant staphylococcal infections require intensive daily bathing (eg, 4% chlorhexidine gluconate, Hibiclens, hibiclens.com; 4% chlorhexidine gluconate with tris-EDTA, TrizCHLOR, dechra-us.com).
Mupirocin ointment can be used on focal areas of infection.
Systemic Antibiotics
Selection of antimicrobial agent is based on culture and susceptibility testing.
Use of vancomycin and linezolid in veterinary patients is controversial because of ethical concerns; these drugs are reserved for treatment of MRSA in humans.
Adverse reactions to potentiated sulfonamides (30 mg/kg PO q12h) and chloramphenicol (50 mg/kg PO q8h) can occur. Clients should be warned of idiosyncratic bone marrow suppression, which rarely occurs with chloramphenicol use in humans.6
MRSA = methicillin-resistant S aureus, MRSIG = methicillin-resistant S intermedius group
Follow-Up
Patient Monitoring
Resolution of the infection is monitored by clinical response. Treatment needs to continue beyond resolution of clinical signs.
After resolution of signs, repeat culture is not recommended. Persistence of MRSIG on carriage sites is common following resolution of the infection (eg, pyoderma).
Complications
Patients receiving potentiated sulfonamide antibiotics optimally should have tear production monitored before treatment is initiated and during treatment if keratoconjunctivitis sicca is suspected.
Chloramphenicol can cause nausea. Drug-induced bone marrow suppression is a rare side effect detected by CBC monitoring.
In General
Relative Cost
Management may be costly when including diagnostics for determining the underlying cause of infection: $$$ to $$$$
Prognosis
Prognosis depends on the site and extent of infection; early intervention in surgical wounds and soft tissue injuries is important.
Multidrug-resistant infections often have a poorer prognosis because of the time and effort required to treat them.
Future Considerations
Minimization or prevention of methicillin resistance will require thoughtful use of antibiotics and consideration of topical agents as first lines of therapy. Treatment based on culture, rather than empirical therapy, is becoming the standard of care.
VAST Recommendations
The Veterinary Antimicrobial Susceptibility Testing (VAST) subcommittee recently made the following recommendations regarding methicillin resistance:
Proposed use of new oxacillin disk diffusion standards (R ≤17 mm) and MIC breakpoints (R ≥0.5 µg/mL) as indicators of mecA-mediated resistance for S pseudintermedius.
The cefoxitin test should not be used for detecting methicillin resistance in S pseudintermedius isolates from dogs. The cefoxitin breakpoints are not predictive of mecA-mediated resistance to methicillin or oxacillin in S pseudintermedius.

Don't Panic
Client education is of utmost importance. Television, internet, and news media have made clients aware (in some instances, frightened) of methicillin resistance. In addition to directing clients to trusted websites (eg, wormsandgermsblog.com) for more information, some key points to stress include:
Antibiotic Gel: Efficacy & Clinical Use
Do not panic
MRSA and MRSIG are not the same
If MRSA is isolated in the pet, then the most likely source is a human
Colonization of humans with MRSIG is transient, if it occurs at all
MRSIG and MRSS have very low zoonotic risk
Wash your hands
At-Home Measures
Hand hygiene (eg, soap and water, alcohol-based hand sanitizers) is the simplest and best way to prevent transmission of infection.
While infection is active, owners should be discouraged from kissing their pet or allowing their pet to lick them.
The pet’s bedding, collars, and clothing should be washed daily or every other day.
During treatment, owners should not allow the pet to sleep in their bed or with children.
Staphylococci can persist in the environment for a short time; routine cleaning can be protective.
Methicillin-Resistant Staphylococcal Infections • Karen A. Moriello
References
1. Veterinary perspective on methicillin-resistant staphylococci. Cohn LA, Middleton JR. J Vet Emerg Crit Car 20:31-45, 2010.2. Methicillin-resistant Staphylococcus aureus and Staphylococcus pseudintermedius in veterinary medicine. Weese JS. Vet Microbiol 140:418-429, 2010.3. Staphylococcal infections. Weese JC. In Greene CE (ed): Infectious Diseases of the Dog and Cat, ed 4—St. Louis: Elsevier Saunders, 2012, p 341.4. Laboratory diagnosis of bacterial infections. Love BC, Jones RL. In Greene CE (ed): Infectious Diseases of the Dog and Cat, ed 4—St. Louis: Elsevier Saunders, 1021, pp 277-283.5. Heteroresistance: A concern of increasing significance? Falagas ME, Makris G, Dimopoulos G, Matthaiou DK. Clin Microbiol Infec_t 14:101-104, 2008.6. Integumentary infections. Ihrke P. In Greene CE (ed): _Infectious Diseases of the Dog and Cat, ed 4—St. Louis: Elsevier Saunders, 2012, p 883.
Suggested Reading
Contamination of pet therapy dogs with MRSA and Clostridium difficile. Weese JS. J Hosp Infect 72:268-269, 2009.Human to dog transmission of methicillin resistant Staphylococcus aureus. Rutland BE, Weese JS, Bolin C, et al. Emerg Infect Dis 15:1328-1330, 2009.Letter to the editor: Proposed changes to Clinical Laboratory Standards Institute interpretive criteria for methicillin-resistant Staphylococcus pseudintermedius isolated from dogs. Papich MG. J Vet Diagn Invest 22:160, 2010.Methicillin-resistant Staphylococcus aureus and Staphylococcus pseudintermedius in veterinary medicine. Weese JS. Vet Microbiol 140:418-429, 2010.Methicillin resistant staphylococci: Implications for small animal practice. Morris D. Dermatology of Companion Animals, CVMA Scientific Presentations: 65-69, 2010.Risk factors for methicillin resistant Staphylococcus aureus (MRSA) infection in dogs and cats: A case control study. Magalhaes RJS, Loeffler A, Lindsay J, et al. Vet Res 41:55-67, 2010.Veterinary perspective on methicillin-resistant staphylococci. Cohn LA, Middleton JR. J Vet Emerg Crit Car 20:31-45, 2010.