Maintaining the Drain
Danielle Browning, LVMT, VTS (Surgery), University of Tennessee, College of Veterinary Medicine
Karen M. Tobias, DVM, MS, DACVS, University of Tennessee
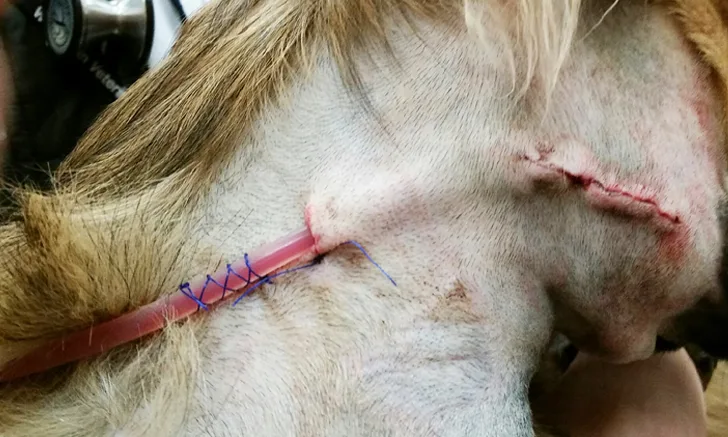
A closed-suction drain exit site is secured with a mattress suture, and a finger-trap suture around the tubing prevents slipping.
After placement of a wound drain, there are specific steps needed to prevent nosocomial infection of the wound and exposure of the team and other patients to infectious material.
Drains are often placed to allow removal of fluid or air, to close dead space, and to prevent seroma formation. On rare occasions, drains may be used to provide medications such as local anesthetics. Drains should always be placed using aseptic technique, and exit sites should always be kept covered.
Figure 2 Adhesive bandage over the exit site of a closed-suction drain

Active drains require a closed system to maintain their suction. After the skin at the exit site has been apposed around the drain with a purse-string or mattress suture (Figure 1), the area can be covered with a small, sterile, nonadhesive dressing and an outer adhesive film or an island dressing (Figure 2).
Passive Drains
Passive drains (eg, Penrose drains, Figure 3) are gravity-dependent and allow fluid, which can be irritating to the periwound skin, to travel around and along the drain and through an exit site in the skin.1 Before bandage application, a thin layer of sterile, petrolatum-based ointment can be applied to the clean, dry skin around (not on) the exit wound to protect the area. Another option is to coat the periwound skin with an acrylate polymer. Once dried, the polymer film will improve dressing adhesion, prevent stripping of the skin when the adhesive is removed, and protect the intact skin from body fluids that can cause maceration and encourage infection.
Figure 3 Penrose drain exiting ventrally on the neck of a dog

A sterile absorptive layer is then placed over the passive drain exit site, including the drain, to collect any exudate. Materials such as cotton, cotton balls, and cast padding should be avoided as the primary contact layer to avoid leaving cotton fibers in the wound.
Additional Bandaging Materials
Options for moderate-to-heavy drainage include gauze surgical sponges, laparotomy pads, polyurethane foams, and super-absorbent pads. Super-absorbent pads are lightweight, less bulky, and more effective at capturing and retaining fluid than traditional dressings.
Low-cost commercial options with similar absorbent functions may include baby or adult diapers, incontinence pads, and feminine hygiene pads. These can all be easily purchased by the owner for home bandage changes.
Figure 4 A soft padded bandage placed over the exit site of the Penrose drain shown in Figure 3.

An absorptive layer of antimicrobial-impregnated gauze (placed against the drain if thick enough to serve as the absorptive layer; otherwise, used with another absorptive dressing) may help prevent growth of opportunistic pathogens. To prevent leaking or contamination, the absorptive dressing should be covered with a semipermeable or nonpermeable outer layer (Figure 4). Clear adhesive drapes or films keep the absorptive layer isolated while permitting visual inspection for strike-through (ie, fluid reaching the outermost layer of the bandage) (Figure 5).
Figure 5 Strike-through of the absorbent gauze is easily seen through the transparent film dressing.

Application Considerations
Before an adhesive layer is placed, the skin must be cleaned, dried, and coated with an acrylate polymer to protect the skin from adhesive stripping. If bandaging an area that is not conducive to a traditional soft padded bandage, another alternative is a tie-over bandage, in which the absorptive and protective layers are held in place by umbilical tape laced through skin suture loops; these can be useful for areas that are difficult to cover or where dressings tend to slip (eg, ventral chest, hip, inguinal, shoulder, midcaudal abdomen of male dogs) (Figure 6).
Figure 6 Tie-over bandage used to secure a closed suction drain placed in a wound on the ventral thorax

Drain molestation by the patient can result in an infection, incisional dehiscence, premature drain removal, or a GI foreign body. A properly fitted Elizabethan collar or similar device is recommended to prevent the patient from chewing its bandage or drain. Body suits (Figure 7), T-shirts, tube socks (cut out leg holes for small dogs and cats), and neck collars can also protect the area and provide a place to secure closed-suction drain tubing.
Figure 7 A body suit and an inflatable neck collar protect the patient from chewing its bandage or drain.

For active drains, bandage change is usually unnecessary until removal unless the patient soils the bandage (eg, with urine, blood, or feces).
For passive drains, bandages are changed before or when strike-through occurs. For patients with resistant infections, bandage changes should occur daily—at the end of the day when possible so that the room can be properly cleaned and sanitized before its next use. Sedation and analgesics may be required for bandage changes, and aseptic technique should always be followed. A clean laboratory coat or gown should be worn and changed after the bandage change is complete. This helps prevent the spread of resistant bacteria. If sterile surgical gloves are not available, examination gloves can be used, but proper hand hygiene is critical. Hands should be washed with soap and water for 20 to 60 seconds or thoroughly coated with an alcohol-based hand sanitizer (>60% alcohol). Hand sanitizers will only suffice if there is no organic debris (eg, dirt, urine, feces, blood) on the hands.
Bandage Removal
The outer bandage layer is cut and removed; if an adhesive drape was used, the drape can be cut just around the absorptive layer, with the rest left intact and attached to the animal as a site for adherence of subsequent drapes. The absorptive layer should be removed and evaluated for drainage quantity and character before disposal in an appropriate trash receptacle. Gloves should be changed and the site around the drain cleaned with an antiseptic solution (eg, 0.05% chlorhexidine or 0.01% hypochlorous acid). Hypochlorous acid wound irrigation solutions have been reported to disrupt biofilms and are effective against drug-resistant bacteria.2 After the area is cleaned, it should be bandaged as previously described.
Drains can incite an inflammatory reaction that can cause fluid production and lead to increased risk for infection. The decision to remove a drain is often based on subjective findings (eg, wound location, drainage characteristics). In general, the fluid should change to serosanguineous and decrease in amount before drain removal. A recent study of closed-suction drains in dogs with clean surgical wounds reported an increased risk for seroma formation after drain removal when the fluid production rate was >4.8 mL/kg/day.3
If fluid drainage does not decrease or character of the fluid does not improve, the wound may have a new or resistant bacterial infection. If infection is suspected, the area should be aseptically prepared and a tissue sample obtained with a blade, scissors, or punch for culture and sensitivity. It may be necessary to open the wound for evaluation, debridement, and topical (open wound) management until culture results are available and the infection has cleared.
Global Note
Readers who want to find veterinary tools mentioned in this article might find them via Jazz Medical, 3M, and K9 Top Coat.