Intrahepatic Splenosis in a Labrador Retriever
Akaterina Davros, DVM, Fort Collins Veterinary Emergency and Rehabilitation Hospital, Fort Collins, Colorado
Kate KuKanich, DVM, PhD, DACVIM (SAIM), Kansas State University
Kelli Almes, DVM, DACVP, Kansas State University
Katherine Tucker-Mohl, VMD, Diagnostic Imaging, P.C., Aurora, Colorado
Megan Wilson, DVM, First Coast Veterinary Specialists, Jacksonville Beach, Florida
Emily Klocke, DVM, DACVS, Kansas State University
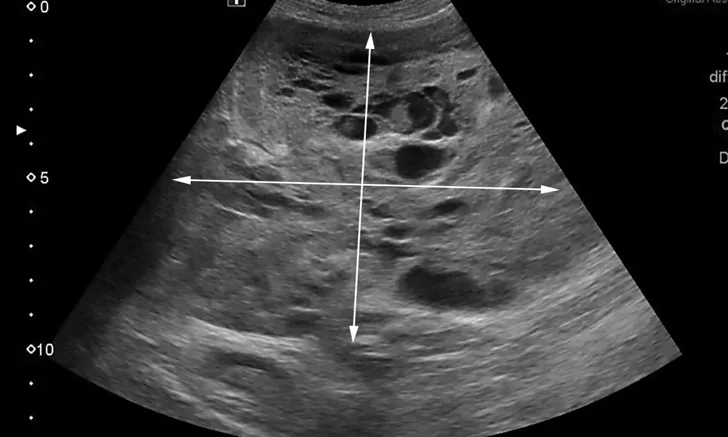
Ultrasonographic appearance of a cavitary mass measuring >10 cm in the left liver on initial presentation. Area of mass demonstrated by arrows
Sophie, a 9-year-old spayed Labrador retriever, was presented to the internal medicine service for abdominal distension. She had a clinical history of acute hemoabdomen, splenectomy, and benign hemangioma on splenic histopathology 2 years prior to presentation. One month prior to presentation, the owners noted that Sophie was gaining weight and that her abdomen appeared larger than usual. Abdominal radiographs obtained by the primary veterinarian revealed a large mid-abdominal mass. She was up-to-date on vaccinations and received routine flea, tick, and heartworm preventives.
Physical Examination Findings
Sophie was bright, alert, and responsive, with normal vital signs. The only abnormal examination findings were abdominal distension and a large and palpable left-sided cranial abdominal mass.
Diagnosis
Mild leukocytosis (17.3 × 103/μL [17.3 × 109/L]; reference range, 4.3-13.6 × 103/μL [4.3-13.6 × 109/L]) and mild regenerative anemia (hematocrit, 33% [reference range, 40%-57%]; reticulocytes, 150 000/μL [150 × 109/L]) were noted. The remainder of the CBC, coagulation profile, and serum chemistry profile were within normal limits. Abdominal ultrasonography demonstrated a large (>10 cm in diameter) hepatic mass (Figure 1) originating from the left aspect of the liver that contained numerous variably sized anechoic regions and mild anechoic peritoneal effusion. Two smaller masses were visible in the hepatic parenchyma (Figure 2). Ultrasound-guided fine-needle aspiration of the large hepatic mass was performed, and cytology showed evidence of previous hemorrhage and mild extramedullary hematopoiesis. Ultrasound-guided biopsies were not considered safe because of the risk for hemorrhage due to the cavitated nature of the masses. The top differential diagnosis was malignant neoplasia (eg, hemangiosarcoma, histiocytic sarcoma, hepatic adenocarcinoma); benign (eg, hemangioma) or infectious (eg, fungal or parasitic) lesions were considered possible but unlikely. Thoracic radiographs were unremarkable.

Ultrasonographic appearance of a second hepatic mass measuring 5.7 cm on initial presentation. Area of mass demonstrated by arrows
TOP DIFFERENTIAL DIAGNOSIS:
MALIGNANT HEPATIC NEOPLASIA
Treatment & Long-Term Management
Although neoplasia was suspected, it could not be confirmed without histopathology. The owners elected for abdominal exploratory surgery to attempt either complete mass resection or debulking with biopsy for histopathology. The owners were prepared to consult with the oncology team following surgery if neoplasia was confirmed. Three liver masses and one mass adhered to the linea alba with omental adhesions were removed, leaving smaller masses and irregularities throughout the liver. The largest mass (11 cm), located in the left medial liver lobe, was firm and vascular and interspersed with pockets of unclotted blood. The left medial liver lobe containing the largest liver mass, 2 additional liver masses, and a mass adhered to the linea alba with omental adhesions were all removed, leaving smaller masses and irregularities throughout the liver. On histopathologic examination, the hepatic masses were found to be composed of red pulp, extramedullary hematopoietic tissue, rare white pulp, and smooth muscle trabeculae (Figure 3). The mass associated with the linea alba was also composed of ectopic splenic tissue, with similar components, consistent with splenosis. No evidence of neoplasia was seen. Recovery was uneventful, and the patient was discharged 2 days postoperatively.

Histopathology of hepatic mass with compressed hepatocytes (star) and splenic red pulp (diamond). Necrosis can be seen in the lower left corner.
Prognosis & Outcome
Abdominal radiographs taken 3 months postoperatively were unremarkable. Sophie experienced a brief episode of vomiting 4 months postoperatively, and packed cell volume (PCV) was 34%; however, ultrasonography was not performed, and she recovered without therapy. Five months postoperatively, she was clinically normal, and a routine recheck identified a PCV of 39%. Ultrasonography at this time identified a 10.5-cm cavitated mass in the right liver near the porta hepatis (Figure 4) and an 8.6-cm hepatic mass with similar appearance to the presurgical ultrasound findings; hemoabdomen was not present. Although recurrence of hepatic splenosis was suspected, hemangiosarcoma or other neoplasia could not be ruled out. Cytology revealed extramedullary hematopoiesis.

Ultrasonographic appearance of 10.5-cm cavitated mass in the right liver lobe near the porta hepatis on recheck examination 5 months postoperatively. The mass was suspected to be a recurrence of hepatic splenosis, and cytology was consistent with extramedullary hematopoiesis; however, biopsies for histopathology were not pursued. Area of mass demonstrated by arrows
Follow-up abdominal ultrasonography and blood work were performed every 2 to 3 months postoperatively. Sophie underwent a unilateral arytenoid lateralization for laryngeal paralysis (11 months after splenosis diagnosis) and a gastropexy after gastric dilatation-volvulus (1 year after splenosis diagnosis), both without complication. She was clinically stable 14 months postoperatively, with a PCV of 36%, no evidence of hemoabdomen or thoracic metastasis, and a stable appearance of the hepatic masses, with the largest mass measured at 9.85 cm. She continued long-term treatment with Yunnan Baiyao (500 mg PO q8h) for potential hemostatic effect.1
Splenosis is a rare complication of splenic trauma or rupture leading to implantation of splenic tissue onto vascular sites in the abdomen or intravascular seeding from surgical splenectomy. Whereas in humans splenosis typically occurs 5 to 10 years after splenectomy,2 in dogs, splenosis has been diagnosed concurrently with a splenic lesion3 or 2 to 5 years after trauma or splenectomy.4-8 Prior to Sophie’s diagnosis, intrahepatic splenosis had been reported in 5 dogs,4-8 and splenosis had been reported in the pancreas,3 mesentery,6,7 abdominal wall,6,7 diaphragm,7 and jejunum6 of dogs. Splenosis can be an incidental finding6 but more often has been reported with inappetence, weight loss, lethargy, weakness, abdominal distension, and/or abdominal pain in dogs.3-5,7,8 Two dogs had hemoabdomen secondary to rupture of intrahepatic splenosis lesions.7,8 Although splenosis is benign in nature, true prognosis and recurrence rate are unknown due to the rarity of this condition. Two of the 5 patients in the literature were euthanized at or around the time of diagnosis, and the remaining 3 cases were reported to have survived at least 5, 8, and 10 months postdiagnosis.4,6,7 Sophie’s survival of at least 14 months (at the time of this article’s publication) provides important prognostic information. In contrast, median survival for hemangiosarcoma after surgery and chemotherapy is 6 months.<sup9 sup>
Intrahepatic Splenosis at a Glance
If an abdominal mass is palpated, abdominal radiography and ultrasonography can help further characterize the mass, but prognosis should not be decided on imaging appearance alone.
Thoracic radiography is recommended to look for pulmonary metastasis prior to laparotomy.
If removing a large vascular mass, the clinician should be prepared to transfuse the patient.
All surgically excised masses should be submitted for histopathologic examination.
Hepatic splenosis is rare, and further prognostic information is needed. Repeating abdominal ultrasonography every 1 to 3 months is suggested to monitor for recurrence.
The Take-Home
Splenosis should be considered a differential diagnosis for hepatic or abdominal masses in dogs with a history of splenic trauma or splenectomy. Not all cavitated hepatic masses in older, large-breed dogs are neoplastic. Histopathology is required to assess whether neoplasia is present so that appropriate treatment options and prognosis can be determined.
PCV = packed cell volume