Eyelid Mass with Conjunctival & Periocular Swelling in a Cat
Sarah Bosch, DVM, Kansas State University
Susan Nelson, DVM, Kansas State University
Jessica Meekins, DVM, MS, DACVO, Kansas State University
Lisa M. Pohlman, DVM, MS, DACVP, Kansas State University
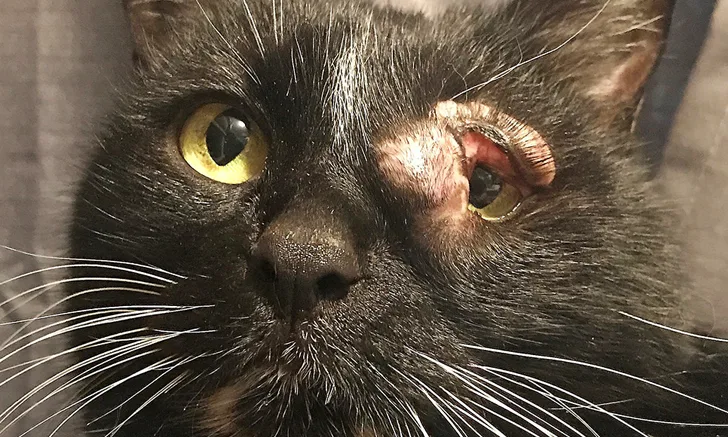
The entire upper eyelid and periocular skin around the medial canthus and extending to the medial lower eyelid associated with the left eye are moderately to severely and diffusely thickened and alopecic. Image courtesy of Guinevere Rava (veterinary student and Belinda’s foster caretaker)
Clinical History & Signalment
Belinda, a 9-lb (4-kg), 5-year-old spayed domestic shorthair cat, was returned to an animal shelter approximately one year after adoption with a recurring mass on the upper left eyelid (Figure 1). The conjunctiva and periocular tissue around the left eye were thickened and inflamed; periocular alopecia was also present. Belinda had been acting normally at home, and the surrendering owner had no other concerns.
An incisional biopsy of the mass had been performed one year prior, and a cutaneous mast cell tumor (MCT) was diagnosed; at the time, histopathology showed the mast cells extended to the surgical margins, which is expected with incisional biopsy, and no mitotic figures were seen.
Physical Examination
On physical examination, Belinda was bright, alert, and responsive. Her vital parameters were within normal limits. The entire upper left eyelid and periocular skin around the medial canthus and extending to the medial lower eyelid exhibited moderate to severe diffuse thickening and alopecia. The left mandibular lymph node was mildly enlarged. The remainder of the physical examination was within normal limits.
Diagnostics
There were no significant abnormalities on CBC and serum chemistry profile. Premedication with diphenhydramine (2 mg/kg IM) was administered prior to fine-needle aspiration of the mass and mandibular lymph node. Cytology of the mass was highly cellular and composed predominantly of variably granulated mast cells (Figure 2). There was mild to moderate anisocytosis and mild anisokaryosis. Binucleated cells were frequent.
Left mandibular lymph node aspirate showed frequent, well-granulated mast cells distributed individually and in groups among a population of lymphocytes, including predominantly small lymphocytes with fewer medium and large lymphocytes and scattered plasma cells (Figures 3 and 4). Given the cytologic findings from the mass and lymph node, abdominal ultrasonography was performed. No abnormalities were seen on ultrasound, but a fine-needle aspirate of the spleen revealed an increased concentration of mast cells scattered individually and in small groups among normal splenic lymphoid and hematopoietic tissue (Figure 5).

FIGURE 2
Fine-needle aspirate of the subcutaneous, periorbital mass showing a sample composed predominantly of variably granulated mast cells. Well-granulated (solid arrows) and poorly granulated (circles) mast cells can be seen. Eosinophils (arrowheads), macrophages (asterisk), and occasional lymphocytes (curved arrow) and spindle cells (dashed arrow) consistent with reactive fibroblasts are also present. No mitotic figures were identified. Modified Wright’s stain, 1,000× magnification
Diagnosis: Feline Cutaneous Mast Cell Tumor With Regional Lymph Node & Splenic Metastasis
Treatment
Surgery was not pursued because of the presence of metastatic disease. Belinda was placed in palliative care in a permanent foster home. She was prescribed diphenhydramine (12.5 mg PO every 12 hours), famotidine (10 mg PO every 24 hours), and prednisolone (7.5 mg PO every 24 hours). Periodic attempts were made to decrease the prednisolone dose; however, increased swelling, erythema, and pruritus with self-injury were consistently noted, and the initial prescribed dose (7.5 mg PO every 24 hours) was determined to be the appropriate maintenance dose.
Prognosis & Outcome
Belinda continued to do well for 15 months, at which time she was returned for evaluation of moderate, focal, cranial abdominal pain with apparent nausea and vomiting. She weighed 9.72 lb (4.41 kg). CBC and serum chemistry profile were unremarkable, and imaging was considered but declined. Maropitant (4.4 mg SC every 24 hours for 3 days), lactated Ringer’s solution (100 mL SC), and buprenorphine (0.044 mg via buccal oral transmucosal route every 8 hours for 3 days) were administered. Famotidine, prednisolone, and diphenhydramine were continued at the previously determined doses. One month later, Belinda was again stable and maintaining a good quality of life.
Treatment at a Glance
Pretreatment with diphenhydramine is recommended prior to MCT aspiration or surgery.9
Surgical removal is the treatment of choice for a solitary cutaneous MCT. Tumor recurrence is low regardless of whether complete surgical excision is obtained.2,3
Histopathology should be performed to determine MI, which is the most significant prognostic indicator for feline cutaneous MCTs.1
Diphenhydramine, famotidine, and prednisolone can be used as medical therapy for feline cutaneous MCTs.9
Consultation with a veterinary medical oncologist to discuss other chemotherapeutic options as necessary is recommended.8,9
Discussion
Cutaneous MCTs are the second most common skin neoplasm in cats. Although most feline cutaneous MCTs are benign, ≈10% are aggressive regardless of histologic type.1 MCTs most frequently arise on the head and neck, followed by the trunk and limbs. There can be a solitary lesion, clusters of lesions, or widespread distribution of lesions ranging from papules and plaques to discrete nodules in the skin or subcutis. Cats may exhibit pruritus, erythema, or edema of the affected area.1
The mean age for development of MCTs is ≈10 years,1 but periorbital MCTs are more common in younger cats.2 There is no sex predisposition. Siamese cats may be more predisposed, especially when young, but MCTs in these patients often regress spontaneously within 24 months.3
MCTs generally exfoliate readily via fine-needle aspiration techniques. Mast cells are large round cells with a centralized purple nucleus often obscured by numerous dark purple granules that fill the abundant cytoplasm. The granules, especially in feline MCTs, can stain poorly with quick stains commonly used in many clinics, making in-clinic diagnosis challenging.1,4
Histologically, feline MCTs are divided into mastocytic (more common) and atypical (less common; previously classified as histiocytic) forms. The mastocytic form is further subdivided into well-differentiated and pleomorphic forms.1 A well-differentiated MCT is typically composed of morphologically normal-appearing mast cells that have minimal anisocytosis and anisokaryosis.1 Mitotic figures can be present but are uncommon. Small lymphocyte clusters can also be present. Spindle cell infiltrates can be seen in MCT aspirates but are less common in feline tumors as compared with canine tumors.1 Mast cells in pleomorphic tumors exhibit more variability (including anisocytosis and anisokaryosis) and eosinophilic infiltrates.1 The cytology of atypical MCTs more closely resembles histiocytes, as opposed to mast cells, and can also contain lymphocytic and eosinophilic infiltrates.1 The cytologic features seen in pleomorphic and atypical MCTs do not correlate with malignant behavior.1
The most important prognostic factor for feline cutaneous MCTs is mitotic index (MI). Low MI (ie, <1 per 10 high-power fields [HPFs]) is associated with a better prognosis. High MI (ie, >5 per 10 HPFs) is associated with a poorer prognosis. Other factors associated with poor prognosis include multiple (>5) simultaneous cutaneous tumors, spread to local lymph nodes, low or moderate cytoplasmic granularity, and a high Ki67 index.1
The treatment of choice for a solitary cutaneous MCT is surgical removal. Studies demonstrate that tumor recurrence is low regardless of whether complete surgical excision is obtained.2,3 Multiple de novo cutaneous MCTs occur in a significant number of cats, and pet owners should be advised of this possibility.1 Additional clinical staging should be considered for all cats with cutaneous MCTs, but cats with any factors for poor prognosis (eg, high mitotic tumor index, >5 simultaneous cutaneous tumors, others as mentioned previously) and cats with visceral MCTs should be fully staged with an evaluation that includes CBC, serum chemistry profile, urinalysis, lymph node aspiration, abdominal ultrasonography with splenic aspiration, thoracic radiography, and possibly bone marrow aspiration. Mastocytemia is more common in cats with MCTs as compared with dogs, and ≈10% of cats with a single cutaneous MCT have mastocytemia.1
Medical treatment for feline cutaneous MCTs may include diphenhydramine (2-4 mg/kg PO every 12 hours), famotidine (1 mg/kg PO every 12 hours), and prednisolone (1-2 mg/kg PO every 24 hours to start).5-7,8 Other potential treatment options may include chemotherapy (eg, lomustine), small molecule inhibitors (eg, imatinib mesylate [tyrosine kinase inhibitor]), and radiation therapy. These therapies may increase survival in some cats; consultation with a veterinary medical oncologist is therefore strongly recommended.8-10
Take-Home Messages
Cutaneous MCTs are the second most common skin neoplasm in cats1 and are generally seen in older cats; however, periorbital MCTs are more common in younger cats.2
Young Siamese cats may be more prone to developing cutaneous MCTs, which can spontaneously regress.3
Feline cutaneous MCTs are usually benign, regardless of histologic type, and recurrence at the site is low regardless of complete surgical excision, although new tumors can occur. Uncommonly, cutaneous MCTs can be aggressive or associated with visceral (splenic or intestinal) disease.1-3
Mast cells exfoliate well with aspiration; however, the granules may stain poorly with quick stains, especially in cats. Cytologic diagnosis should be confirmed by a clinical pathologist.1,4
MI is the most important prognostic indicator in feline cutaneous MCTs; high MI (>5 per 10 HPFs) is associated with a poorer prognosis.1
Clinical staging should ideally include CBC, serum chemistry profile, urinalysis, lymph node aspiration, abdominal ultrasonography with splenic aspiration, thoracic radiography, and possibly bone marrow aspiration.1
Up to 10% of cats with a single cutaneous MCT can have circulating mast cells, making CBC with blood smear review the minimum necessary diagnostic recommendation prior to surgery.1