Diagnosing & Treating Babesiosis in Dogs
Adam J. Birkenheuer, DVM, PhD, DACVIM, North Carolina State University
Michael F. Di Cicco, DVM, North Carolina State University
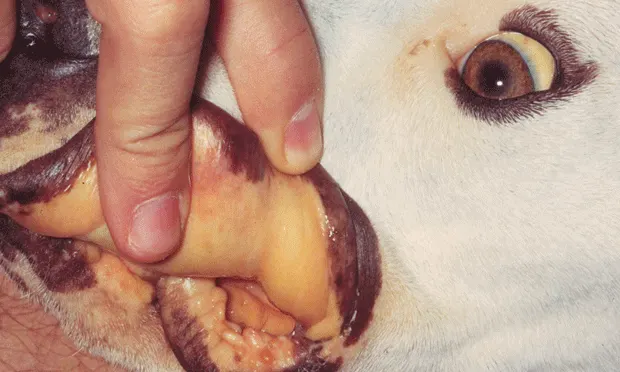
Canine babesiosis, an important worldwide tick-borne protozoal disease, is characterized by hemolytic anemia, thrombocytopenia, febrile illness, and splenomegaly.
Profile
Definition
Clinical disease in dogs is variable and can be sublinical, chronic, or life threatening.
Babesia spp can vary with geographic location and can share common features, but the virulence and pathophysiology also vary with each species.
Historically, canine Babesia spp have been divided into two categories—large and small—based on intraerythrocytic form.
Molecular testing has allowed further characterization by genotype into several subspecies.
Molecular characterization is superior to morphologic characterization for predicting relationships between organisms.
Hematologic, immune, and lymphatic systems are most commonly affected, but the nervous and urinary systems may also be involved.
Signalment
Infections can occur in any canine breed at any age.
B gibsoni infections are more prevalent in American pit bull terriers.
B canis vogeli infections are more prevalent in greyhounds.
Risk Factors
Tick infestation or exposure (transmission time from tick to the host is unknown).
Recent dog bite (B gibsoni).
Blood transfusion from infected donor.
Splenectomy.
Immunosuppression.
Transplacental transmission.
Geographic Distribution
Small Babesia spp
B gibsoni: worldwide
B conradae: southern California only
B microti-like (B annae, Theileria annae): Spain; also prevalent in North American foxes; a single case reported in a dog (Mississippi)
Large Babesia spp
B canis vogeli: worldwide
B canis canis: Europe
B canis rossi: South Africa
B coco: United States (in splenectomized or immunosuppressed dogs)
Pathophysiology
Hemolytic anemia caused by Babesia spp is multifactorial, including immune-mediated destruction (intra- and extravascular), direct parasitic injury, and subsequent oxidative stress.
Thrombocytopenia is a result of immune-mediated destruction or a consumptive process secondary to endothelial injury.
Signs
History
Thorough history should include:
Signalment
Environmental exposure
Travel history
Flea and tick prevention history
History of recent dog bites or blood transfusions
Physical Examination
Clinical signs can vary substantially because of differences among Babesia spp and individual patient response to infection.
Patients can present with either acute or chronic illness.
Typical findings include:
Lethargy
Pale mucous membranes
Splenomegaly
Waxing and waning pyrexia
Bounding pulses
Lymphadenopathy
Generalized weakness
Jaundice
Vomiting (more commonly reported with B conradae infection)

Canine blood smear of Babesia canis showing paired large merozoites (pair tear-drop forms).

Canine blood smear of Babesia gibsoni showing single merozoites (signet-ring form).
Diagnosis
Definitive Diagnosis
Light microscopy:
High specificity
If intracellular parasite is identified by experienced hematologist, the patient can typically be said to be infected. However, microscopic examination has poor specificity for predicting genotype of the parasite.
Low sensitivity.
Capillary blood may enhance.
Commercially available quick stains work well.
Large Babesia spp
3–7 µm long
Single or pair tear-drop forms
Small Babesia spp
1–3 µm long
Signet-ring form
Indirect fluorescent antibody (IFA) testing:
Cannot differentiate among Babesia spp.
Titers of ≥1:64 support exposure.
False-negative results possible with peracute or acute disease or infection with a Babesia species or strain that differs from the laboratory species or strain.
Polymerase chain reaction (PCR) testing:
High specificity and sensitivity.
Can determine species or subspecies with specific PCR assay or DNA sequencing.
False-negative results are possible with low numbers of circulating parasites.
Sensitivity can be increased by performing 2 or 3 consecutive tests 2–4 weeks apart.
False-negative results are also possible if primers are “too specific” (ie, will only amplify B gibsoni and will not detect B canis).
Differential Diagnosis
Immune-mediated hemolytic anemia, immune-mediated thrombocytopenia, zinc toxicity, rickettsial diseases, bartonellosis, leptospirosis, dirofilariasis with caval syndrome, systemic lupus erythematosus, and neoplasia.
Laboratory Findings
CBC and direct blood smear:
Thrombocytopenia is the most common feature regardless of the Babesia spp. Macrocytic anemia and autoagglutination are variable (not all animals are anemic).
Leukogram is highly variable.
Pronounced leukemoid response to intraerythrocyte parasites, reticulocytosis, and leukopenia are possible.
Serum biochemistry profile: hyperglobulinemia, hyperbilirubinemia, increased liver enzyme activities, azotemia (B canis rossi, B gibsoni, B annae), and hypoalbuminemia.
Urinalysis: bilirubinuria, hemoglobinuria, and proteinuria.
Coombs test (direct): can be positive in 85% of cases.
Coagulation testing:
Thrombocytopenia.
Disseminated intravascular coagulation has also been reported.
Infectious disease titers to rule out coinfection or other infection:
Ehrlichia spp, Rickettsia rickettsii, Bartonella spp, Anaplasma spp.
Radiography:
Abdominal radiographs to rule out metallic/zinc foreign body.
Thoracic radiographs can identify neoplastic process.
Other Diagnostics
Molecular genetic detection includes broad range of Babesia PCR, multiplex PCR (to rule out coinfection), and DNA sequencing.
Treatment
Treatment choices largely depend on the Babesia species identified.
Most dogs show response to treatment in 24–72 hours; however, it can take up to 7 days before results are apparent.
Inpatient or Outpatient
Hospitalization may be required, but many dogs can be treated as outpatients.
Medical
Supportive care and therapy largely depend on clinical presentation and patient assessment.
Markedly anemic dogs may require blood transfusion (component therapy with packed RBC and fresh whole blood is ideal).
While not available in the United States, hemoglobin-based oxygen-carrying solutions have successfully alleviated clinical signs in anemic dogs with babesiosis.
Decision to transfuse should be based on animal’s history, clinical signs, and hematologic abnormalities.
Physical examination abnormalities (eg, tachycardia, tachypnea, bounding or water-hammer pulses, generalized weakness, collapse) can indicate need for RBC transfusion.
In general, transfusion is indicated with hematocrit concentration of ≤15% and clinical signs consistent with anemia.
Medications
Drugs/Fluids
IV fluids for correction of dehydration and hypovolemia.
Imidocarb dipropionate (6.6 mg/kg IM once, repeat in 7–14 days) reduces morbidity and mortality in most cases of Babesia spp infection.
Treatment of choice for B canis vogeli but is ineffective for clearance of B gibsoni and B conradae.
Pretreatment with atropine (0.02 mg/kg SC 30 minutes before imidocarb) reduces cholinergic side effects (ie, salivation, lacrimation, vomiting, diarrhea, tachycardia, dyspnea).
Diminazene aceturate (3.5–7 mg/kg SC or IM q1–2wk) is effective against B canis but is unavailable in the United States.
Not capable of clearing B gibsoni or B conradae infection.
Atovaquone (13.3 mg/kg PO q8h) and azithromycin (10 mg/kg PO q24h) combination therapy has effectively cleared B gibsoni and B conradae infections.
Atovaquone should be given as liquid suspension with a fatty meal to ensure adequate absorption.
Clindamycin (25 mg/kg PO q12h), metronidazole (15 mg/kg PO q12h), and doxycycline (5 mg/kg PO q12h) have been associated with clearance of B gibsoni after administration for ~3 months, but true treatment efficacy is unknown.
Precautions
Use of glucocorticoids or other immunosuppressive drugs for immune-driven hemolysis in canine babesiosis is controversial.
Use of immunosuppressive drugs during treatment may predispose patient to other infections, reduce reticuloendothelial system clearance of the organism (with possible increase in parasitemia), and impede complete clearance of the infection.
The authors do not recommend immunosuppressive drugs for treatment of babesiosis.
Follow-Up
Patient Monitoring
In hospital settings, hematocrit concentration and platelet count can be monitored daily until improvement is seen.
Continue monitoring q1-2wk until hematocrit and platelet numbers have normalized.
PCR testing at 60 and 90 days after treatment is recommended to rule out treatment failure.
Serology is not recommended posttreatment, as titers do not necessarily wane after treatment.
If the patient fails to respond favorably to therapy, additional screening for coinfection should be considered.
Complications
At high doses, imidocarb dipropionate and diminazene aceturate have been associated with liver and kidney failure.
Prevention
Vector control is the primary means of preventing infection.
Dog’s skin and hair coat should be examined frequently for ticks, especially in known endemic areas.
All ticks should be removed within 24 hours.
Use of topical acaricides and environmental control may minimize transmission of tick-borne infections.
Use of 0.9% amitraz-impregnated collar, fipronil (S-methoprene) + amitraz, and imidacloprid 10% with permethrin 50% has been associated with reduced transmission of Babesia spp.
Proper infectious disease screening of donor before blood donation can prevent transmission via transfusion.
All animals should be screened before placement in a kennel or boarding facility.
Vaccine for B canis canis is available in Europe.
Reduces severity of disease but does not prevent infection.
Does not confer protection against B canis vogeli, B canis rossi, or B gibsoni.
Another European vaccine combines antigens from B canis canis and B canis rossi.
Reduces severity of disease but does not prevent infection.
In General
Relative Cost
Management and treatment of canine babesiosis can be costly, especially if hospitalization for administration of blood products and supportive care is required.
Prognosis
In general, prognosis is good with early diagnosis and appropriate treatment.
Infection that is not cleared may remain subclinical for life.
Persistently infected dogs have potential for relapse, especially following splenectomy or if immunosuppressed.
Client Education
Transmission to dogs primarily occurs through arthropod infestation, blood contamination, or a bite wound.
Canine babesiosis is not believed to be zoonotic.
Take-Home Points
Babesiosis can occur in any canine breed at any age.
A good history and thorough examination are key.
Diagnostics include light microscopy (poor sensitivity), IFA testing (unable to distinguish Babesia spp), and PCR assay (high specificity and sensitivity).
Treatment approaches depend on the Babesia spp identified.
Preventive measures include vector control and screening animals before placement in a kennel or boarding facility.