Common Pulmonary Diseases in Cats
Douglas Palma, DVM, DACVIM (SAIM), The Animal Medical Center, New York, New York
What is important to understand about the differential diagnoses for primary bronchial disease and pulmonary disease in cats?
Most feline patients presented with respiratory signs have primary bronchial disease. However, pulmonary disease does occur in cats.
Congestive Heart Failure
Congestive heart failure (CHF) is a common acute cause of parenchymal changes characterized by interstitial to alveolar pulmonary infiltrates. CHF in cats is distinct from dogs in its clinical features and radiographic findings.
CHF is characterized by a relatively acute onset of respiratory signs. Increased respiratory effort, respiratory rate, lethargy, and inappetence are primary complaints. Coughing is rarely exhibited in these patients.
Observation of breathing may reveal dyspnea with mixed inspiratory and/or expiratory effort with pulmonary edema. Severe disease may result in orthopneic posturing and failure to maintain recumbency. A restrictive pattern characterized by short, shallow breathing with increased inspiratory effort is usually noted with pleural effusion. Cats with pleural effusion frequently adopt sternal posturing and may exhibit a subtle overall change in breathing pattern. Unlike dogs, pleural effusion is common in cats with CHF and may delay immediate recognition of signs.
Physical examination may identify changes in intensity of bronchovesicular sounds, generation of abnormal sounds (eg, wheezing, crackles), and alterations in breathing pattern or body posturing. Auscultation of the heart may reveal a heart murmur, gallop rhythm, sinus tachycardia, and/or other arrhythmias but may also be normal. Many cats with CHF are hypothermic. Rarely, cyanosis of mucous membranes and lingual surfaces may be noted.
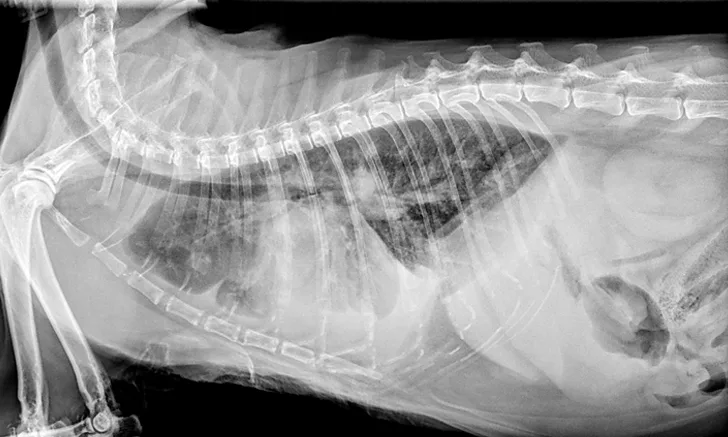
Overt cardiomegaly may be understated on radiographs because of changes in internal diameter that do not affect the overall cardiac silhouette. Vertebral heart score >9.3 supports a CHF diagnosis.1 Pulmonary venous congestion can be absent, and pulmonary arterial distension may be appreciated. The presence of pleural effusion with simultaneous pulmonary infiltrates is supportive of CHF (Figure 1).2
Pulmonary parenchymal infiltrates vary widely in appearance and distribution. Radiographic pulmonary changes may include lobar to multifocal alveolar disease, patchy pseudonodular appearance, peribronchial pattern, and diffuse, unstructured interstitial disease (Figure 2). Echocardiography may support a diagnosis (left atrium dimension, >15.7 mm; left atrial aortic root ratio, >1.4; E-wave velocities, >1.2).2,3

FIGURE 1A
Congestive heart failure. Note the pleural effusion (arrows).
Heartworm Disease
In endemic regions, heartworm should be suspected in patients with compatible clinical signs. Heartworm-associated respiratory disease (HARD) may be noted with immature infections, whereas mature infections may cause disease via local residence of adult worms in the pulmonary arteries. Clinical signs are often subtle but may include persistent tachypnea, intermittent coughing, and increased respiratory rate. Additional clinical signs include anorexia, weight loss, emesis, neurologic manifestations, and sudden death.4
Radiographs may reveal a diffuse bronchointerstial pattern, pulmonary arterial enlargement, pulmonary hyperinflation, and/or right-sided cardiomegaly. However, these findings are nonspecific and may represent other conditions.
Serologic testing is recommended for immature infections, as antigen testing is invariably negative and echocardiography is uncommonly confirmatory. Serology may be supportive of infection but could indicate exposure alone.
Bronchopneumonia
Bronchopneumonia is less common in cats than in dogs but may be more common in kittens. In the author's experience, it occurs more commonly with pre-existing bronchial disease and/or administration of corticosteroids. In addition, historical upper respiratory tract disease and nasal discharge are common.5
Pneumonia is commonly associated with systemic signs (eg, lethargy, hiding, inappetence). Clinical signs include dyspnea, nasal discharge, and coughing.6 A leukocytosis, regenerative left shift, and/or pyrexia may be suggestive but are often absent.7
Radiographic evaluation is variable. Classic cranioventral distribution is less common in cats (Figure 3). A bronchial pattern is most common; however, bronchointerstitial, alveolar, and more structured pseudonodular patterns can occur.6,8 Occasionally, pulmonary abscesses may be appreciated and resemble pulmonary neoplasia (Figure 4).

Radiograph of bronchopneumonia with more classic cranioventral distribution (arrows). Other diseases, including CHF, have a similar distribution. Lack of this pattern does not rule out infection.

Radiograph of pulmonary abscessation. The pulmonary nodules (arrows) could be mistaken for pulmonary neoplasia. The margins of these nodules are slightly less well demarcated than in neoplasia. Improvement was achieved with antibiotic therapy.
Additional diagnostic testing, including airway sample acquisition, to eliminate other differential diagnoses should be based on clinical signs. Mycoplasma spp is the most commonly identified etiologic agent; special media should be collected in addition to routine aerobic cultures.8
Lungworms
Aelurostrongylus abstrusus is the most common lungworm in cats worldwide. Infection is most commonly acquired through ingestion of paratenic hosts. Although many cases are subclinical, ingestion of a large number of larvae may cause respiratory signs characterized by chronic coughing, dyspnea, and debilitation.9 Eosinophilia is sporadically observed.
Thoracic radiographs may reveal a bronchointerstitial pattern, alveolar infiltrates, a diffuse bronchial pattern, and (rarely) pleural effusion (Figure 5).10,11 Diagnosis may be made via identification of first-stage larvae from airway cytology specimens or fecal samples. In clinical practice, false-negative fecal samples have been appreciated due to intermittent fecal shedding. Repeated testing may increase sensitivity but has not been evaluated. Therefore, any patient suspected of potential lungworm should be treated. Less common lungworms have been reported in cats, including Paragonimus spp12-14 and Capillaria spp.15

FIGURE 5A
Radiograph of aelurostrongylus abstrusus infection (lungworm). Note the diffuse bronchial pattern (orange arrows), patchy poorly defined soft-tissue nodules (red arrows), and right middle lung lobe alveolar disease (yellow arrow). This pattern could be confused with feline asthma in this case; however, the right middle lung does not appear atelectatic but instead infiltrated. Fine-needle aspiration of the right middle lung lobe confirmed larvae.
Toxoplasmosis
Toxoplasmosis is a multisystemic disease involving the liver, eyes, lungs, neurologic system, musculoskeletal system, pancreas, and/or heart.16 Therefore, clinical suspicion should be increased in patients with multiorgan involvement.
Clinical toxoplasmosis is relatively uncommon and typically occurs in young cats. The most common postnatal infections include predominant respiratory infection or a multisystemic disease process with respiratory involvement. The most common clinical signs include anorexia, lethargy, and dyspnea; however, a myriad of signs related to other organs (eg, nervous system, pancreas, eyes) are possible.16
Radiographic evaluation may reveal a diffuse interstitial to alveolar pattern with mottled lobar distribution. Pleural effusion may be noted in some animals. Tachyzoites may be detected on pulmonary fine-needle aspiration, on bronchial/tracheal cytology, and/or in pleural effusion.17Additionally, tachyzoites may be appreciated in the blood, CSF, and peritoneal effusion. Abdominal imaging may reveal pancreatitis, lymphadenopathy, intestinal masses, and hepatomegaly.
Serologic testing for IgM antibodies is more likely to correlate with clinical disease. A combination of compatible clinical signs, serologic testing results, and response to therapeutic intervention supports a diagnosis of clinical toxoplasmosis.18,19
Additional Multisystemic Infections & Disorders
Uncommon causes of disseminated infections with involvement of the pulmonary parenchyma can occur in cats (see Reported Pulmonary Infections). Clinical suspicion should increase when a cat exhibits systemic signs of illness and multiorgan involvement and when other, more common differential diagnoses have been eliminated. Absence of significant changes on CBC or serum chemistry profile typically does not rule out disease in these cases.
Reported Pulmonary Infections20-31,43
Fungal disease: Histoplasma capsulatum, Blastomyces dermatitidis, Coccidioides immitis, Cryptococcus neoformans, Cryptococcus gattii, Aspergillus fumigatus
Atypical bacterial infections: Mycobacterium microti, Yersinia pestis, Rhodococcus equi, Bergeyella (formerly Weeksella) zoohelcum, Capnocytophaga cynodegmi, Chlamydophila felis
Viral infections: Feline infectious peritonitis (Figure 6)
Protozoal infections: Cytauxzoon felis

Radiograph of atypical distribution of pulmonary feline infectious peritonitis (red arrows). Larger granulomas mimicking neoplasia and pleural effusion may be noted in some cases. Necropsy confirmed pyogranulomatous inflammation and associated coronavirus antigen.
These infections may be associated with a wide degree of radiographic patterns, including unstructured interstitial, structured interstitial (eg, nodular, miliary, masses), or alveolar. Tracheobronchial lymphadenopathy and pleural effusion may be noted in some of these cases. In other cases, pulmonary involvement may not be appreciated on thoracic radiographs.
Pulmonary embolism rarely occurs in cats and is generally associated with a hypercoagulable state. In the author's experience, the most commonly reported concurrent conditions include cardiac disease, neoplasia, corticosteroid administration, disseminated intravascular coagulation, and pancreatitis. Acute respiratory signs in a patient with an underlying condition should raise clinical suspicion.32,33
Idiopathic pulmonary fibrosis has been sporadically reported in cats. This interstitial lung disease has been associated with respiratory distress, tachypnea, and/or coughing. Nonspecific signs have been described. Radiographic findings vary with a diffuse or patchy interstitial, bronchial, or alveolar pattern. Clinical suspicion should increase for patients showing mild cytologic airway changes but severe clinical signs, refractory bronchial disease, or diffuse interstitial or alveolar disease. Definitive diagnosis requires lung biopsy.34,35
Lipid pneumonia may be appreciated in cats secondary to exogenous inhalation of oil (eg, mineral oil) or endogenous generation secondary to obstructive pulmonary disease (eg, bronchial disease, neoplasia). Radiographic features vary widely and can include diffuse interstitial, bronchointerstitial, or nodular patterns (Figure 7). Pleural effusion may be noted. These findings may represent the underlying obstructive disease.36,37
Lung lobe torsion is an uncommon cause of respiratory distress in cats. It may occur spontaneously or secondary to pleural effusion and should be suspected in cats with pleural effusion and parenchymal disease.38-40
Pulmonary neoplasia is often incidental and noted as a soft-tissue opacity in the caudal lung lobes. Bronchial compression may result in coughing, whereas diffuse/metastatic disease may cause systemic signs of illness.41,42 Pulmonary metastatic disease may be subtle in some cases, with a more prominent bronchial pattern in cats (Figure 8).
Atelectasis
Atelectasis, most commonly of the right middle lobe, may be noted incidentally in cats. A mediastinal shift may or may not be present on radiographs and may be mistaken for primary parenchymal disease (Figure 9). Atelectasis may occur secondary to bronchial obstruction; therefore, investigation for active bronchial disease may be indicated if clinical signs are suggestive.

FIGURE 7
Endogenous lipid pneumonia confirmed on lung biopsy (orange arrow, biopsy site). The origin of the lipid pneumonia could not be identified in this case. Note the patchy unstructured interstitial opacities (red arrows).