Sarcoma in Animals and Humans
Sarah Boston, DVM, DVSc, DACVS, ACVS Founding Fellow of Surgical Oncology, ACVS Founding Fellow of Oral & Maxillofacial Surgery, VCA Canada, Newmarket, Ontario, Canada
Andre R. Spiguel, MD, University of Florida

Editor's Note (2019): An earlier version of this article incorrectly stated that ileectomy was performed. The article has been corrected.
This column co-presents clinical cases in a side-by-side format, with the patient—human or veterinary—as the primary variable. It is the hope of the Clinicians Brief team that readers will see the inherent commonalities in managing cases of similar diseases, regardless of species.
The Zoobiquity series is based on Zoobiquity by Barbara Natterson-Horowitz and Kathryn Bowers.
Zoobiquity: Osteosarcoma in a Dog
History
A 10-year-old spayed rottweiler with a 2-week history of left pelvic limb lameness and a palpable mass over the left ilium was referred after fine-needle aspiration findings were consistent with a mesenchymal sarcoma, most likely osteosarcoma.
Clinical Signs
On physical examination, the patient continued to show mild left pelvic limb lameness. There was a 10-to-12cm hard palpable mass overlying the left ilium. The physical examination was otherwise unremarkable.
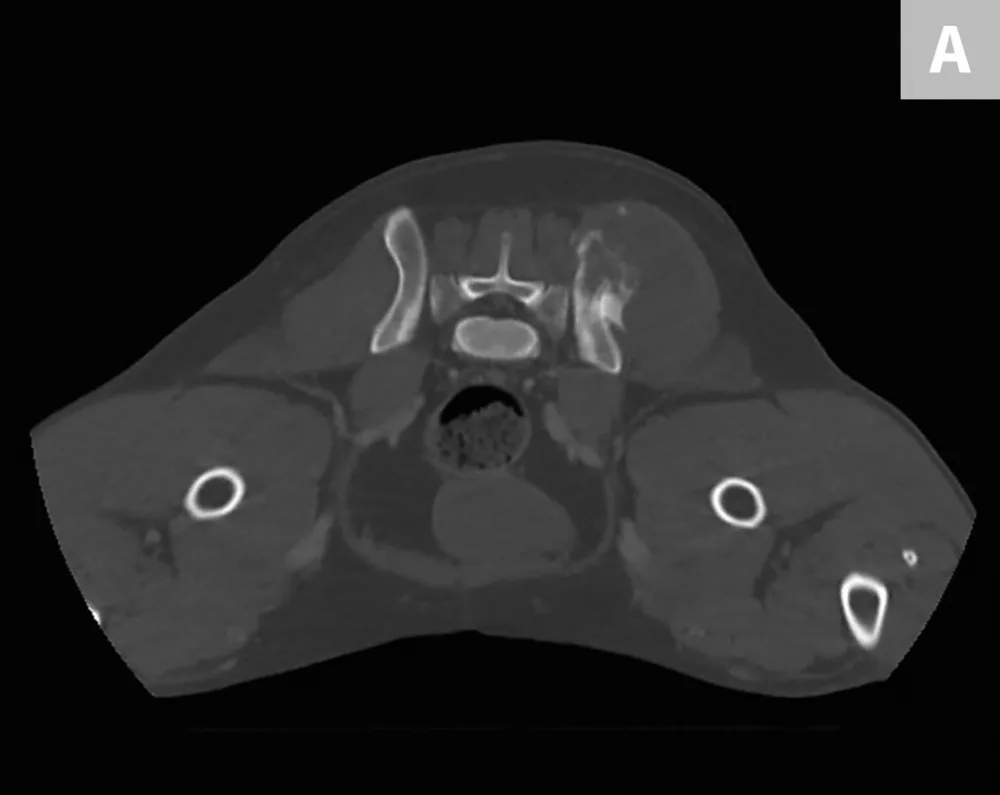
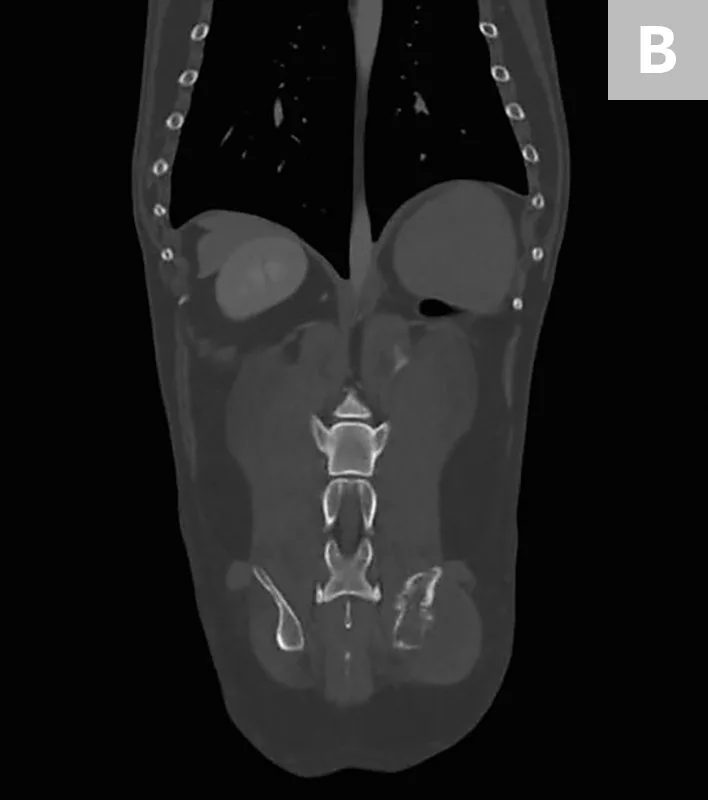
Axial (A) and coronal (B) CT images from the patient. An aggressive bone lesion arising from the left ilium, with extension to the surrounding soft tissues, can be seen.
Diagnosis
CBC, serum chemistry profile, and urinalysis results were unremarkable. Three-view thoracic radiographs showed no evidence of gross metastatic disease. A CT scan of the chest, abdomen, and pelvis showed an aggressive bone lesion of the left ilium, compatible with osteosarcoma, with tumor extension to the surrounding soft tissue (Figure 1). A full-body bone scan was performed. Static images were obtained 2 hours after intravenous injection of Tc99m-MDP. There was marked uptake in the area of the primary tumor (ie, the left ilium). There was also a possible metastatic lesion in the left costal cartilages; however, careful review of the CT scan showed that the increased uptake of radiopharmaceutical at that site was most likely consistent with old trauma rather than metastatic disease (Figure 2).
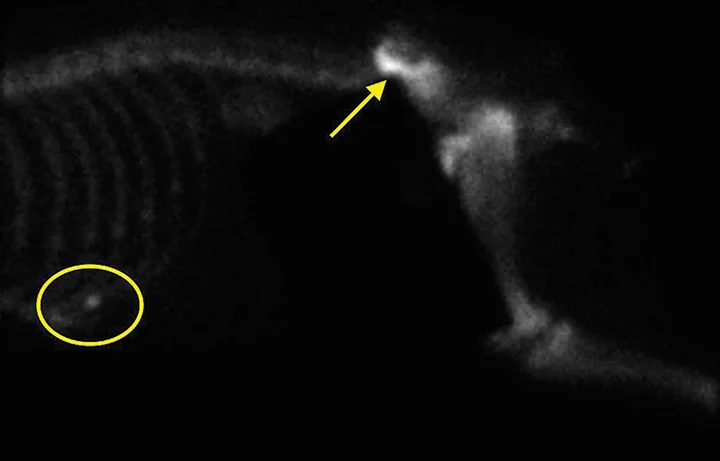
Bone scan image from the patient. Increased uptake of radiopharmaceutical in the left ilium (arrow) and a potential lesion in the left costal cartilage (oval) can be noted.
Treatment
Left iliectomy and stereotactic radiosurgery were suggested to provide curative-intent local control of the tumor. Palliative radiation was also discussed. The owners elected iliectomy and preservation of the left pelvic limb; the left ilium was resected via sacroiliac disarticulation (Figure 3), and the left acetabulum and left pelvic limb were preserved. Postoperative radiographs showed adequate tumor removal (Figure 4).
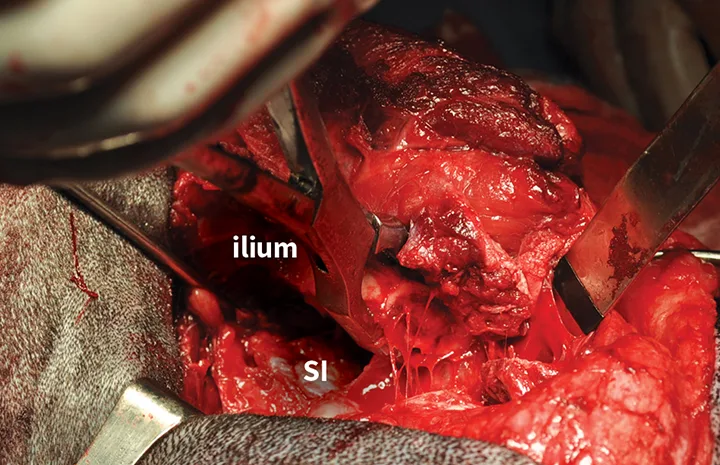
Intraoperative image of iliectomy. The osteotomized ilium is grasped with bone-holding forceps after disarticulation of the sacroiliac joint. The exposed left sacroiliac joint is visible at the level of the sacrum (SI).
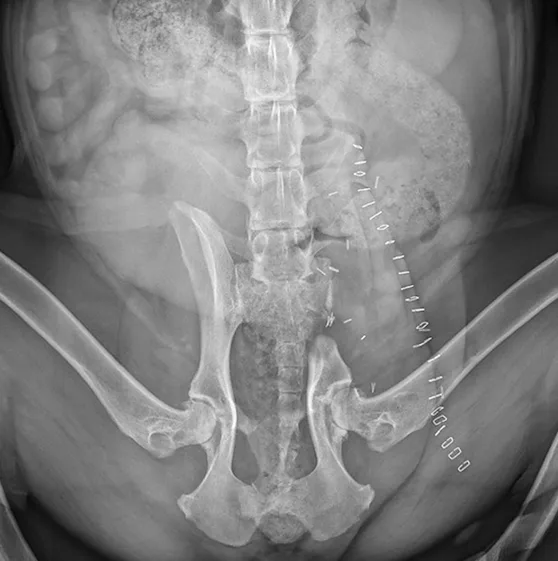
Ventrodorsal postoperative radiograph showing the removal of the affected portion of the left ilium
Outcome
The patient recovered well from surgery in the intensive care unit. She was managed postoperatively with fentanyl (2-5 g/kg/hr CRI) and meloxicam (0.1 mg/kg IV q24h). The dog was walking well with minimal support within 24 hours and was discharged 48 hours postoperatively on meloxicam (0.1 mg/kg PO q24h) and tramadol (3 mg/kg PO q8-12h).
The specimen was inked to assess the margins and submitted for histopathology. Histopathology confirmed a diagnosis of osteosarcoma. The mitotic index was 32 mitotic figures per 10 high-power (400) fields, with moderate-to-marked anisocytosis and anisokaryosis. The margins of excision were complete. Chemotherapy with single-agent carboplatin (6 doses at 300 mg/m2 IV) was initiated and was well tolerated without complication. At 136 days postsurgery, the patient showed no evidence of local recurrence or metastatic disease on pelvic and thoracic radiographs, respectively, and was ambulating well (see Video).
Video of the patient ambulating with minimal support one day postsurgery
Pelvic Chondrosarcoma & Hemipelvectomy in a Man
HISTORY
A 36-year-old otherwise healthy man with a history of multiple hereditary exostoses (MHE) noticed a painful mass over the right ilium that had slowly enlarged over the past year. He was referred to a musculoskeletal oncologist and presented with previously obtained radiographs.
CLINICAL SIGNS
On physical examination, the patient was pleasant and in no acute distress. He had a large nonmobile mass over the right hemipelvis. He was able to ambulate normally without any deficits. The rest of the examination was otherwise unremarkable.
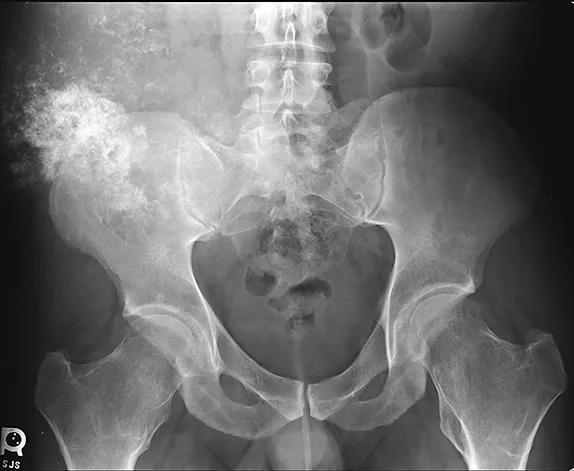
Anteroposterior radiograph showing a large mineralized mass with poorly defined margins and a wide zone of transition arising from the right ilium
DIAGNOSIS
Previously obtained anteroposterior pelvic radiographs (Figure 1) showed a large, poorly defined, mineralized lesion involving the right ilium. Because of the patients physical examination results, history of MHE, and radiographic findings, a secondary chondrosarcoma of the right ilium was suspected. Further diagnostic evaluation, including a CT scan (Figure 2) and MRI of the pelvis, were obtained to further characterize the lesion and begin surgical planning.
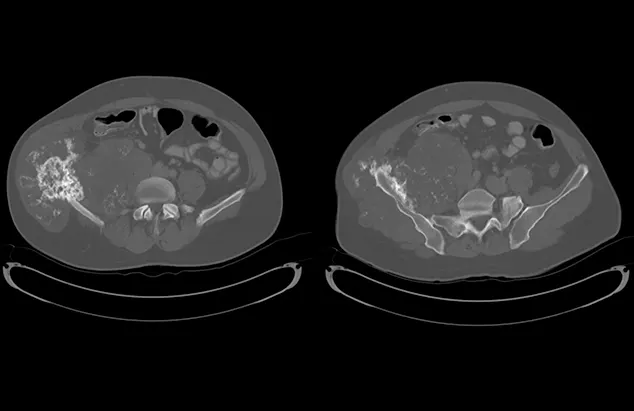
Axial CT images showing a large mass arising from the right ilium with a significant soft-tissue component in both the inner and outer tables of the pelvis
Because the lungs are the most common site of metastasis in cases of chondrosarcoma, staging was determined via bone scan and chest CT, which confirmed that there were no other sites of disease. Staging was determined to be Enneking Stage IB; prognosis was good overall. Core needle biopsy confirmed low-grade chondrosarcoma.
Treatment
Low-grade secondary chondrosarcoma with no other sites of disease is treated definitively with resection when possible. Neoadjuvant treatments (eg, radiation, chemotherapy) are of limited value and used only in palliation with limited success.1-3 Because of the patients age and good prognosis, and no need for adjuvant treatment if a negative margin could be achieved, reconstruction was elected to restore the pelvic ring in the setting of an iatrogenic disruption. The right ilium was resected by cutting through the sciatic notch toward the anterior inferior iliac spine and cutting through the sacral ala posteriorly to ensure a negative margin (ie, internal hemipelvectomy), which allowed for preservation of the right acetabulum and right hindquarter (Figures 3 and 4). Reconstruction was performed using lumbopelvic fixation and a double-barrel vascularized free fibula autograft harvested from the ipsilateral leg (Figure 5). Postoperative radiographs showed resection and reconstruction (Figure 6).
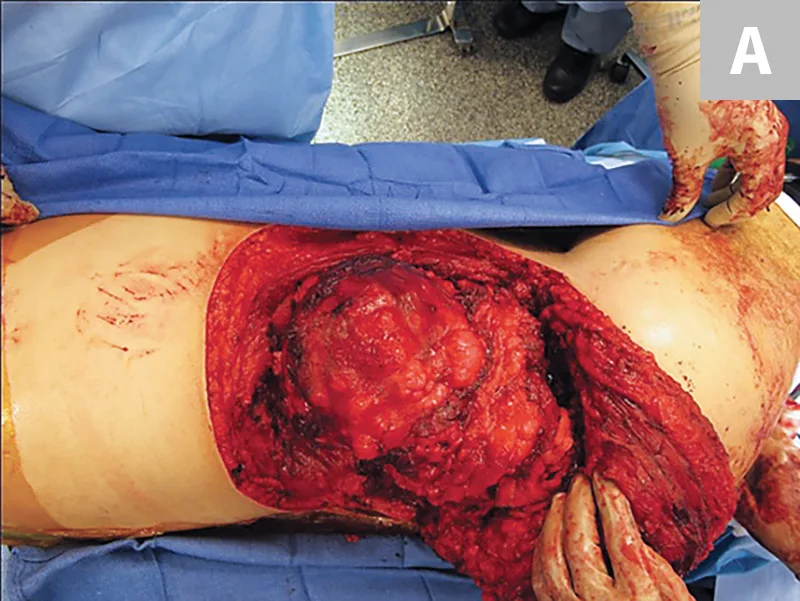
FIGURE 3 Intraoperative photos obtained before bony cuts. The soft tissues are mobilized circumferentially around the mass (A). The microvascular surgeon can be seen harvesting the autograft fibula from the ipsilateral leg while the resection is being performed (B).
OUTCOME
The patient recovered well from surgery. Pain was controlled using a standard lumbar epidural immediately postoperatively. Two days postoperatively, the patient was doing well and the epidural was weaned. Through reconstruction and stabilization of the pelvic ring, the patients recovery was significantly advanced. He was discharged 5 days after surgery on oxycodone (10 mg PO q4-6h as needed for pain) and low-molecular weight heparin for thromboprophylaxis. To protect his construct, he was made toe-touch weight bearing for 3 months.
The patient was seen for surveillance, including radiographs of the pelvis and a CT scan of the chest, every 3 months for 2 years. At 3 years postoperation, surveillance is performed every 4 months, then every 6 months for years 4 and 5 postoperation. After 5 years postoperation, he is considered cured. Because of his history of MHE and increased risk for subsequent secondary chondrosarcomas arising from prior osteochondromas, he will continue to be seen every year for clinical examination, pelvic radiographs, and any imaging of painful or enlarging osteochondromas.