Cardiopulmonary Resuscitation
Melissa Java, VMD, DACVECC, Veterinary Specialty & Emergency Center, Levittown, Pennsylvania
Bridget MacDonald, CVT, VTS (ECC), Veterinary Specialty & Emergency Center, Levittown, Pennsylvania
The RECOVER guidelines have changed! Review the 2024 updates to CPR protocol in this overview.
Definition & Pathophysiology
Cardiopulmonary arrest (CPA) occurs when there is cessation of cardiac function and there are no spontaneous respirations.
CPA can cause global hypoxemia, multiple organ failure, and, eventually, death.1
Cardiopulmonary resuscitation (CPR) provides ventilatory and circulatory support to patients that undergo CPA.
The goal is to establish oxygen delivery—most importantly, to the cerebrum and myocardium—without causing further organ injury.1
Return of spontaneous circulation (ROSC) occurs when there is a sustained perfusing rhythm, significant breaths (more than an agonal breath), and more than a brief pulse.2
Published in 2012, the Reassessment Campaign on Veterinary Resuscitation (RECOVER) guidelines are the first evidence-based, consensus-based guidelines for veterinary CPR.3
Causes
Causes of CPA are often multifactorial. Common causes include4-6:
Cardiac disease
Pulmonary disease
Upper airway disease
Polytrauma
Sedation or anesthesia
Toxin ingestion
CNS disease
Other major organ dysfunction
Risk Factors
Ill, compromised, intoxicated, traumatized, anesthetized, and/or sedated patients are at risk.
Patients at increased risk include those that are critically ill, debilitated, and have more comorbidities present.
Common risk factors
Hypoxemia
Hypercarbia
Cardiac arrhythmias
Severe anemia
Hypothermia
Hypotension
Acid-base disturbances (eg, severe metabolic or respiratory acidosis)
Electrolyte disturbances (eg, hyperkalemia, hypocalcemia, hypoglycemia)
Vagal stimulation
Critical illness
Pre-existing disease
Sedation or anesthesia
Clinical Signs of Impending Arrest
Changes in respiratory rate or pattern, most notably agonal breaths
Bradycardia
Hypotension
Arrhythmias
Hypothermia
Cyanosis
Pupillary dilation
Diagnosis
A patient undergoes CPA when heart function stops and spontaneous breathing ceases.
The CPA patient is unresponsive with no palpable pulse, heartbeat, or spontaneous respirations.
A patient can undergo respiratory arrest and can have a heartbeat but no spontaneous respirations.
If no intervention (intubation and positive pressure ventilation) is taken, full CPA may ensue if heart function ceases.
Basic Life Support
Basic Life Support (BLS) is the cornerstone of CPR and includes recognizing the ABCs (airway, breathing, circulation).
Previous human CPR guidelines recommended performing the ABCs in this order.
Recent human CPR guidelines have moved in favor of CABs (ie, circulation, airway, breathing).7
Veterinary CPR follows the RECOVER3 guidelines, which recommend performing chest compressions and intubation simultaneously in cases for which there are multiple rescuers involved or cases of witnessed CPA due to primary cardiac disease.
If there is a single rescuer and it is an unwitnessed CPA or if there is a single rescuer and CPA is not due to primary cardiac disease, then RECOVER guidelines state that traditional ABCs (intubation/ventilation before chest compressions) can be started provided there is not a significant delay in chest compressions.
It is recommended that patients be intubated while in lateral recumbency so chest compressions are not delayed while the patient is being intubated.
If the patient has just undergone respiratory arrest but still has a heartbeat, the patient should be immediately intubated and ventilated.
Although no studies exist on mouth-to-snout ventilation, the RECOVER guidelines recommend using this technique if intubation is not possible; it is recommended to alternate 30 chest compressions with 2 breaths when using this technique.3
Chest Compressions
As soon as CPA is recognized, chest compressions should be performed at a rate of 100 to 120 per minute.
In most cases, lateral recumbency is the preferred position (either left or right is acceptable).
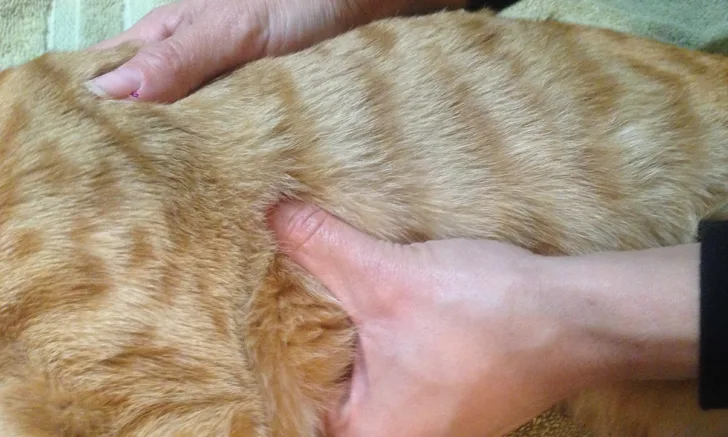
In most cats and small dogs (<10 kg), compressions can be performed directly over the heart (point of maximal intensity, usually between the 4th-5th intercostal space) to maximize blood flow (called the cardiac pump theory in which direct compression of the heart causes blood flow).
Circumferential 1-hand placement can be used; one hand is used to stabilize the back of the animal, and the other compresses directly over the heart using the index and middle fingers and thumb (Figure 1).

FIGURE 1 In circumferential 1-hand placement, one hand is used to stabilize the back of the animal. The other hand compresses directly over the heart using the index and middle fingers and thumb.
Alternatively, a 2-hand technique can be used in larger cats and small dogs that do not have normal chest compliance or in situations in which the compressor fatigues using the 1-hand technique.
In this technique, the hands are stacked one on top of the other in parallel, and the compressor uses the palms of the hands to compress directly over the heart (Figure 2).

FIGURE 2 In a 2-hand technique (used in larger cats and small dogs without normal chest compliance or in situations in which the compressor fatigues using the 1-hand technique), the hands are stacked one on top of the other in parallel, and the compressor uses the palms of the hands to compress directly over the heart.
In larger animals (medium- to giant-breed dogs), it is recommended to compress the widest part of the thorax using the palms of both hands stacked one on top of the other in parallel, with the animal in left or right lateral recumbency.
Called the thoracic pump theory, this maximizes blood flow; compressing the widest part of the thorax causes the greatest intrathoracic pressure change, which then compresses the heart to cause blood to flow.
In dogs with deep, narrow chests (eg, greyhounds), it is reasonable to compress directly over the heart in lateral recumbency to maximize blood flow via the cardiac pump theory.
In barrel-chested animals (eg, brachycephalic breeds), it is reasonable to perform chest compressions directly over the heart with the patient in dorsal recumbency to provide blood flow via the cardiac pump theory.
The chest should be compressed about one-third to one-half its width, allowing for complete recoil in between compressions so as not to inhibit venous return.
Changing the individual performing compressions every 2 minutes is strongly recommended to minimize fatigue and provide the most effective chest compressions.
Interruptions in chest compressions are strongly recommended to be minimized to <10 seconds every 2-minute cycle.
Intubation
Intubation should be performed in lateral recumbency to allow simultaneous compressions.
Intubation should be performed using a laryngoscope.
Intubation may be difficult, especially if trauma has occurred.
Other useful techniques include using a stylet and direct digital palpation.
If intubation is not possible, an emergency tracheotomy should be performed.
After intubation, the tube should be secured and the cuff inflated.
Positive-pressure ventilation should be initiated with 100% oxygen at a rate of 10 breaths/minute.
A resuscitator bag or anesthesia machine can be used; it is important that the gas inhalant is turned off and the system is flushed well.
The clinician should avoid hyperventilating the patient; this can cause an increase in intrathoracic pressure that can decrease venous return and lead to a worse outcome.
Abdominal Compression
Interposed abdominal compression may be performed.
This is a more challenging technique that requires more team members and involves alternating abdominal compressions with chest compressions to optimize venous return.
Advanced Life Support
Advanced Life Support (ALS), which expands and complements BLS, includes specific therapies based on each individual arrest situation:
Electrocardiogram (ECG) monitoring
Administration of drugs
Electrical defibrillation
Cardiac pacing
ECG Monitoring
ECG evaluation should be performed as soon as possible.
Common arrest rhythms include:
Pulseless electrical activity (PEA)
Asystole
Ventricular fibrillation (VFib)
Pulseless ventricular tachycardia (pulseless VTach)
In cats and dogs, the most common arrest rhythms are PEA and asystole, followed by VFib and pulseless VTach.6,8
VFib is the most common arrest rhythm in humans.9
Drug Administration
An easy-to-read CPR drug-dosing chart with the names of the drugs and dosages (volumes provided according to body weight to prevent calculation errors) is available in the RECOVER guidelines3 (posters available at VECCS.org).
The preferred route of drug administration is IV, ideally through a centrally placed catheter or central line to optimize myocardial drug delivery.
If a central IV catheter is not available, a peripheral IV catheter is the next preferred route; a forelimb is preferable to a hindlimb.
Using intraosseus catheters is the next-preferred route of drug administration if IV is not possible.
These are particularly useful in small or neonatal patients.
All drugs and fluids can be administered via this route.
When no other routes are available, intratracheal administration may be used.
The dose of the drug administered should be at least doubled, diluted with sterile saline or water.
After drug administration, a couple of breaths should be allowed to disperse the drug through the pulmonary circulation.
All drugs except sodium bicarbonate and calcium gluconate can be administered this way.
Because of risk for complications, direct intracardiac administration of drugs is strongly discouraged except possibly during open-chest CPR.10
The 2 vasopressors used during CPR include epinephrine and vasopressin.
Epinephrine is a catecholaminergic vasopressor used for its α-1 adrenergic effects, which can cause peripheral vasoconstriction during CPR; the β-adrenergic effects, which can cause an increase in heart rate and contractility, may be detrimental during CPR.
There is both a low dose (0.01-0.02 mg/kg) and a high dose (0.1-0.2 mg/kg).
It is recommended to start with the low dose and administer every 3 to 5 minutes every other BLS cycle (ie, 3-5 mins of ventilation/chest compressions); if no ROSC occurs after prolonged CPR, the high dose can be used.
Vasopressin is a noncatecholaminergic vasopressor that causes peripheral vasoconstriction via specific V1 vasopressin receptors located on smooth-muscle vasculature.
Vasopressin may be useful because of its lack of negative β-adrenergic effects, as compared to epinephrine.
Vasopressin may be more effective than epinephrine in an acidic environment, which occurs during a prolonged arrest.
The dose of vasopressin is 0.8 U/kg.
The RECOVER guidelines3 recommend vasopressin in place of epinephrine every 3 to 5 minutes or in addition to epinephrine every 3 to 5 minutes.
Atropine, an anticholinergic drug used to abolish parasympathetic tone, is indicated in vagally mediated arrests or those that develop a sinus bradycardia.
The dose is 0.04 mg/kg IV, IO, or IT.
RECOVER guidelines3 state that atropine can be used routinely during CPR given the lack of detrimental effects. It may be most useful in cases of asystole and PEA associated with high vagal tone.
If hypocalcemia- or hyperkalemia-induced cardiac arrest occur, calcium gluconate should be considered.
If anesthetic-related arrests have occurred, antagonists should be considered:
0.01 mg/kg flumazenil (benzodiazepine antagonist)
0.04 mg/kg naloxone (opioid antagonist)
0.1-0.2 mg/kg atipamezole or yohimbine (α-2 antagonists)
Inhalant anesthetics should be discontinued and the breathing circuit flushed well with oxygen. IV fluids should be considered in hypovolemic patients. Fluid administration in euvolemic patients may decrease cerebral and coronary perfusion and may lead to poorer outcomes.
Defibrillation
Defibrillation is the only effective treatment for VFib or pulseless ventricular tachycardia.
The initial shock dose using a biphasic defibrillator is 2-4 J/kg (if externally defibrillating) or 0.2-0.4 J/kg (if internally defibrillating). The RECOVER guidelines3 state that biphasic is preferred over monophasic.
If using a monophasic defibrillator, the dose is 4-6 J/kg (if externally defibrillating) or 0.4-0.6 J/kg (if internally defibrillating).
Previous human guidelines recommended 3 consecutive shocks, but this is no longer recommended.
The CPR drug-dosing chart in the RECOVER guidelines3 has defibrillation dosing available according to body weight so that the correct joules can be quickly set to deliver.
If the animal has been in VFib for <4 minutes, give 1 immediate shock followed by BLS for 2 minutes before evaluating the ECG.
If the animal has been in VFib for >4 minutes, perform BLS for 2 minutes before administering a shock.
Before evaluating the ECG, BLS should be performed for 2 minutes after delivering the shock.
If the rhythm has not converted, another shock is recommended at a 50% increase in the current followed by 2 minutes of BLS.3
If a defibrillator is not available, a precordial thump (ie, a single, sharp blow over the precordium) may be administered.
If defibrillation is ineffective, amiodarone or lidocaine can be tried.
Amiodarone is a Class III antiarrhythmic; the dose is 5 mg/kg IV.
Lidocaine, a Class IB antiarrhythmic, is known to increase defibrillation threshold and should only be tried if defibrillation is ineffective and amiodarone is not available. The dose is 2 mg/kg IV in dogs.
Open-Chest CPR
Open-chest CPR should be considered when there is significant intrathoracic disease (eg, pericardial effusion, penetrating chest wounds/chest wall trauma, pleural space disease).
Open-chest CPR requires a large, skilled team; advanced aggressive post-cardiac arrest care; and high financial responsibility for the client.
CPR Monitoring
The most common ways to monitor CPR efforts include ECG analysis, palpation of peripheral pulses, and monitoring end-tidal CO2 (ETCO2).
ETCO2, the gold standard, is related to cardiac output when ventilation is constant.
Higher ETCO2 (>15-20 mm Hg) values during CPR may correlate with ROSC.
ETCO2 is also useful for verifying endotracheal tube placement during intubation as the ETCO2 will always be higher in the trachea than in the esophagus.
Follow up
ROSC occurs in about 35% to 45% of animals, but only 5% to 10% will survive to discharge.4,11
ROSC is thus only an intermediate step.
Most CPA patients quickly undergo another arrest if the underlying cause is not quickly identified and treated.
68% of dogs and 37.5% of cats undergo another arrest.5
These patients may undergo a post-cardiac arrest syndrome that involves multiple-organ dysfunction and contributes to mortality.
Quickly finding and reversing the underlying cause is critical.
Additional monitoring and treatment is necessary to decrease further organ injury.
Appropriate management should be performed in an intensive care unit.
Additional Testing
Additional testing once ROSC occurs includes:
Frequent physical examinations
Evaluation of blood pressure
ECG, packed cell volume, total solids, and blood glucose
Blood gas, lactate levels
Electrolyte analysis
Pulse oximetry
End-tidal capnography
Monitoring urine output12
Depending on the patient’s clinical disease, other necessary tests may include:
CBC
Serum chemistry profile
Blood typing
Coagulation profile
Urinalysis
Cultures (eg, blood, urine)
Radiography
Ultrasonography
Prognosis
The prognosis for animals undergoing CPA is grave.
The prognosis for survival to discharge for cats that undergo CPA in a hospital setting is 9.6%. For dogs that undergo CPA in a hospital setting, the prognosis for survival to discharge is 4.1%.4
The cause of arrest is important in determining prognosis.
Animals that have a reversible cause (eg, anesthesia, drug-related event), witnessed event, or that undergo respiratory arrest alone have a much better prognosis.5-6,11,13
Client Education
It is important to guide owners in choosing a resuscitation code (ie, do not resuscitate, closed chest CPR, open-chest CPR) appropriate for the patient’s clinical condition.
If a patient has a terminal condition, it is important to tell owners that it may not be in the pet’s best interest to resuscitate.