Abdominal Pain & Pyrexia in a Cairn Terrier
Ariadna Ribas Latre, DVM, DECVIM, MRCVS, Dick White Referrals, Six Mile Bottom, Cambridgeshire
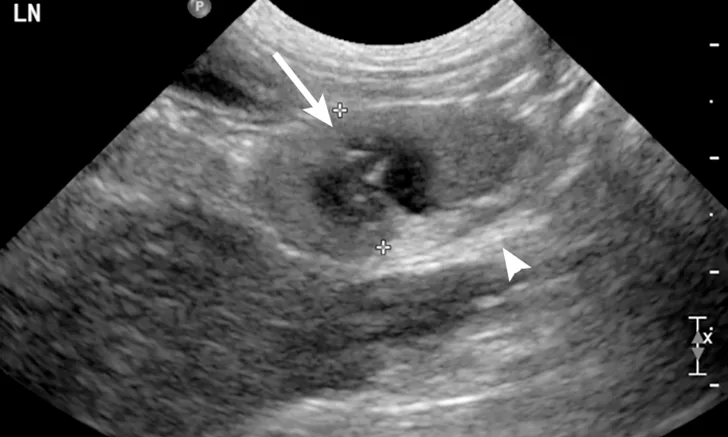
Ultrasound image of a jejunal lymph node. A moderately enlarged lymph node with a rounded heterogeneous appearance and ill-defined hypoechoic patches can be seen (arrow). The mesenteric fat around the lymph node was diffusely hyperechoic (arrowhead).
Ruby, a 3-year-old, 15.6-lb (7.1-kg) spayed cairn terrier, was presented for a 24- to 36-hour history of hyporexia and abdominal pain. She had no previous medical conditions, vaccinations and flea and tick preventives were up to date, and she had not traveled outside the United Kingdom. Initial assessment revealed pyrexia (rectal temperature, 103.8°F [39.9°C]). She was started on potentiated amoxicillin (20 mg/kg SC) and referred to a specialty clinic for further evaluation.
Physical Examination
On presentation, Ruby was lethargic but alert and responsive. Abdominal pain, pyrexia, and hyporexia were of main concern. Her BCS was 5/9, her rectal temperature was 104.4°F (40.2°C), and she was ≈7% dehydrated. She demonstrated apparent generalized discomfort on abdominal palpation. No peripheral lymphadenopathy was noted, and the rest of the physical examination was unremarkable.
Diagnostics
Differential diagnoses for abdominal pain should include any infectious, inflammatory, or neoplastic disease that affects the abdominal organs; rupture, volvulus, or obstruction of an abdominal organ; and referred pain from other muscular, neurologic, or bony structures. Differential diagnoses for pyrexia should include drug administration; toxin ingestion; and infectious, inflammatory/immune-mediated, and neoplastic disease. Differential diagnoses for hyporexia are broad and can be related to abdominal pain or pyrexia; however, other causes may include primary GI or systemic disease; local disease affecting structures involved in prehension, mastication, or swallowing (ie, oral, dental, muscular, bone, neurologic conditions); pain; and behavior.1
CBC and serum chemistry profile revealed a moderate neutrophilic leukocytosis (23,300/μL; reference interval, 3,000-11,500/μL) without toxic changes. The remaining CBC and serum chemistry results were within normal limits. C-reactive protein (CRP) levels were also assessed for systemic inflammation, and levels were markedly elevated at 29.4 mg/dL (reference interval, <1).
Abdominal ultrasonography revealed the presence of multiple enlarged, heterogeneous, and rounded cranial mesenteric lymph nodes (Figures 1, above, and 2). These findings were suggestive of round cell neoplasia, lymphadenitis (infectious or noninfectious), or reactive lymphadenopathy. There was a small amount of free abdominal fluid, which was sampled and submitted for analysis; this was compatible with a nonseptic suppurative exudate. Culture results of the fluid were negative. Fine-needle aspirates and cytology of the abdominal lymph nodes were consistent with neutrophilic lymphadenitis (Figure 3). No infectious agents were seen. The rest of the abdominal ultrasound was unremarkable.

Ultrasound image of a jejunal lymph node. A moderately enlarged lymph node with a lobulated heterogeneous appearance and ill-defined hypoechoic patches can be seen (arrows). The mesenteric fat around the lymph node was diffusely hyperechoic (arrowhead).

Cytology of a fine-needle aspirate from a jejunal lymph node revealing large numbers of variably degenerate neutrophils (arrowheads). No bacteria were observed. Small numbers of macrophages (arrow) and small and medium lymphocytes (stars) were present. These changes are compatible with neutrophilic lymphadenitis. Magnification 100×
Differential diagnoses for neutrophilic lymphadenitis include infectious disease, immune-mediated disease, neoplasia, and sterile lymphadenitis.2 Thoracic radiography was performed to assess for any distant cause of infection, neoplasia, or inflammation, but the results were unremarkable. Urinalysis was unremarkable, urine culture was negative, and serology for arthropod-borne disease (eg, Ehrlichia spp, Anaplasma spp, Borrelia spp) was negative.
Ultrasound-guided trucut biopsies of the abdominal lymph nodes were submitted for histopathology and culture to exclude infectious lymphadenitis. Histopathology confirmed neutrophilic lymphadenitis (Figure 4), and lymph node tissue cultures (bacterial and fungal) were negative.

Hematoxylin and eosin stained histologic image of a trucut biopsy from a jejunal lymph node. Normal lymph node architecture was effaced by the presence of sheets of degenerate neutrophils (black arrows) and smaller numbers of macrophages (arrowheads) and plasma cells (white arrows). Magnification 40×
Diagnosis: Sterile Steroid-Responsive Neutrophilic Lymphadenitis
Treatment
Ruby was initially treated with IV crystalloid fluid therapy for dehydration and opioid pain relief (methadone at 0.2 mg/kg every 4 hours) for abdominal discomfort. Antibiotic therapy with potentiated amoxicillin was continued.
Because an infectious cause was not found, treatment with glucocorticoids was started (prednisolone at 1.5 mg/kg every 24 hours). Ruby remained normothermic 48 hours after treatment was initiated; she had a good appetite and there was no evidence of abdominal pain. She was discharged, and treatment with glucocorticoids was continued.
Recheck examinations were scheduled to occur every 3 to 4 weeks. Ruby remained clinically well, and her CRP levels normalized (0.4 mg/dL; reference interval, <1). Repeat CBC also revealed resolution of the inflammatory leukogram. Glucocorticoid treatment was gradually decreased by 25% to 30% every 3 to 4 weeks, on condition that clinical signs remained resolved and physical examination was normal. Abdominal ultrasonography was performed 1 month after initial diagnosis, revealing resolution of the mesenteric lymphadenopathy.
Treatment at a Glance
Glucocorticoid treatment can be started after infectious causes have been excluded.
Most patients seem to respond to glucocorticoid therapy, and rapid resolution of clinical signs is associated with resolution of lymphadenopathy.
Immunosuppressive glucocorticoids are often necessary to control clinical signs.
Other immunosuppressive medications may be needed if patient response is suboptimal or steroid adverse effects are severe.
Prognosis & Outcome
Treatment with glucocorticoids was discontinued ≈4 months after diagnosis because the patient remained clinically well. Ruby remained healthy without treatment 2.5 years after initial presentation.
Discussion
Sterile steroid-responsive lymphadenitis (SSRL) is not well-characterized in dogs; however, it should be considered in dogs with pyrexia of unknown origin and inflammatory lymphadenopathy for which no underlying cause can be found.
There is little information available about SSRL. Two retrospective, multicentric studies discussed this condition (including clinical signs, diagnostic approach, and treatment) in different breeds,3 particularly in English springer spaniels.4 Other case reports/series have also reported on SSRL, especially in English springer spaniels with or without concurrent dermatologic signs.5-8
A diagnosis of SSRL can be determined via cytology and/or histopathology by documenting an inflammatory infiltrate in lymph nodes without evidence of infectious agents or other underlying disease. It is key that infectious causes be excluded before SSRL is diagnosed; geographic variations are important considerations in these cases, as prevalence of infectious disease can vary depending on the region or country. Good clinical response is initially seen in most patients after glucocorticoid initiation, but other immunosuppressants can be considered if patient response is suboptimal or steroid adverse effects are severe. Relapses are possible after discontinuation of therapy, so monitoring may be necessary.3,4 CRP evaluation has been used as a monitoring technique. Young female dogs, particularly English springer spaniels, seem to be predisposed to lymphadenitis.3,4 Previous reports of lymphadenitis in English springer spaniels included a case of sterile neutrophilic-macrophagic lymphadenitis associated with nodular panniculitis,6 granulomatous necrotizing lymphadenitis,7 and mineral-associated lymphadenopathy.8
SSRL should be on the differential diagnosis list for young adult dogs presented with pyrexia and lymphadenopathy.
Take-Home Messages
SSRL should be considered in dogs that have pyrexia and variable degrees of lymphadenopathy.
Clinical signs can vary.
Definitive diagnosis of SSRL involves extensive investigation to rule out detectable underlying infectious, inflammatory, or neoplastic causes.
Lymphadenopathy can be external, internal, or both.
Young female dogs, particularly English springer spaniels, seem to be predisposed.3,4
CRP can be used for monitoring purposes.
Relapses are possible.
CRP = C-reactive protein, SSRL = Sterile steroid-responsive lymphadenitis