Top 5 Oncologic Conditions that Respond to Surgery in Dogs
Sarah Boston, DVM, DVSc, DACVS, ACVS Founding Fellow of Surgical Oncology, ACVS Founding Fellow of Oral & Maxillofacial Surgery, VCA Canada, Newmarket, Ontario, Canada
In most cases, tumor type and stage should be diagnosed before surgical intervention to determine the most appropriate treatment plan. This information, combined with the owner’s goals for therapy, can help determine the appropriate surgical approach.
The following are 5 common oncologic conditions that respond to surgery.
1. Soft-Tissue Sarcoma
Soft-tissue sarcoma (STS) is the disease most often used as an example in explaining the basic principles of the surgical management of cancer. In many cases, sarcomas can be cured with surgery, particularly if they are low-to-intermediate grade and if surgical oncology principles are followed. This means always having knowledge of the tumor type before excision with wide margins. Wide margins should include 3-cm radial margins and 1 fascial plane deep.1 A fascial plane is a barrier to tumor cell invasion, and it is necessary to remove this deep fascial plane to ensure that deep invading cells are removed. A fascial plane may include fascia, a layer of muscle, or bone. Subcutaneous tissue, fat, and tumor pseudocapsule are not an adequate deep barrier for tumor-cell invasion.
If adequate margins cannot be achieved with a wide excision of STS, consideration should be given for marginal or radical excision. Radical excision is the removal of an entire anatomical segment (eg, limb amputation). Many antebrachial STS can be successfully removed without amputation by employing wide excision with antebrachial fascia as the deep barrier. This defect can be reconstructed with a free skin graft or another skin-reconstruction technique2 (Figure 1). If the mass is overlying the carpus or elbow, a marginal excision should be considered. In low- and intermediate-grade tumors and geriatric patients, this may be sufficient for long-term control.3 For high-grade tumors (III), consideration should be given to follow up with radiation therapy.4


(A) Antebrachial soft-tissue sarcoma that is treated with wide resection. Note the deep layer of antebrachial fascia that serves as the deep margin and a barrier to tumor cell invasion (arrow). The surgeon’s fingers to the right of the arrow are between the extensors of the antebrachium and the fascial layer that is deep to the tumor. (B) Reconstruction of defect after antebrachial soft-tissue sarcoma wide resection with a free skin graft
2. Osteosarcoma
Osteosarcoma (OSA) is the most common primary bone tumor in dogs.5 Unfortunately, most cases are not cured with surgery because of the high risk for metastatic disease.5 Patients should be staged for gross metastasis to lung or another bone before surgery (Figure 2). Staging methods vary depending on clinician preference, but 3-view thoracic radiographs and/or thoracic CT and bone scan are recommended.6 Limb amputation is the most commonly performed surgical treatment; major benefits include removal with wide margins of the tumor, removal of the source of pain, and removal of potential for ongoing metastasis.



(A) Radiograph of a distal radial osteosarcoma. Centered at the distal metaphysis of the radius, there is evidence of permeative and moth-eaten lysis. There is also concurrent ill-defined periosteal proliferation consistent with an aggressive bone lesion. (B) CT scan of distal radial osteosarcoma in the same patient. Coronal view of limb in bone window. There is more evidence of geographic bone lysis and evidence of periosteal proliferation. Consistent with an aggressive bone lesion. (C) Bone scan of the distal radial osteosarcoma in the same patient. Note the increased uptake of radiopharmaceutical in the distal radius of the patient (arrow).
In some cases, another surgical option may be limb salvage, which is most commonly performed in the distal radius and involves the surgical removal of the affected bone. The defect is most commonly bridged using an endoprosthesis (ie, a metal implant with a bone plate extending from the metacarpals to the proximal radius). Although the limb is usually preserved, the rate of complications (eg, infection, local recurrence, implant failure) is extremely high.7 Limb salvage should only be considered when amputation is not an option for medical reasons (eg, a patient with significant concurrent orthopedic or neurological disorders). There are cases in which a client will not accept amputation and limb salvage is considered; client education in these cases is critical.
3. Mast Cell Tumor
Mast cell tumors (MCTs) can be reliably diagnosed by microscopic examination of a properly stained fine-needle aspirate. Because MCTs vary in appearance, it is important to aspirate all masses. For example, masses can look like nonhealing wounds; soft subcutaneous masses with a lipoma-like appearance; or more classically, an inflamed dermal mass. Once a diagnosis is reached, the next step is to decide whether to pursue full staging before wide excision; this includes aspiration and/or removal of regional lymph nodes and abdominal ultrasound with or without aspiration of the spleen and liver. MCTs rarely metastasize to the lungs at initial presentation; however, thoracic radiographs are often included as baseline staging and to assess the intrathoracic lymph nodes.
A recent study showed that the local lymph node is sentinel to distant metastasis and that in the face of a negative regional lymph node, distant spread is considered unlikely.8 If the mass is relatively small and can be easily removed with wide margins, lymph nodes should be assessed as a minimum and full staging should be considered.
In other cases for which the morbidity of surgery is high or the MCT has gross characteristics of malignancy (eg, large size, ulceration, firm adherence to underlying tissues), full staging should be considered before surgical removal. High-grade MCTs should be fully staged. Wide excision is the recommended therapy for MCTs (Figure 3). Surgical treatment is the mainstay of MCT treatment, but adjuvant chemotherapy may be indicated in high-grade tumors and cases with evidence of metastasis. Adjuvant radiation therapy may be indicated in cases in which local control cannot be achieved with surgery alone.


(A) MCT over thorax, planned wide resection (B) Specimen after wide resection. The specimen will be inked on the radial and deep margin for tumor-margin assessment.
Some dogs are predisposed to developing multiple MCTs. In these cases, it does not appear that their prognosis is worse if each mass is treated like an individual event and removed appropriately.9
The classic grading scheme for MCTs is grade I, II, or III.10 The dilemma is that most tumors are grade II, and it can be challenging to know how they might behave. In addition, there is documented inconsistency within and between pathologists regarding the 3-grade system.11 A new grading system categorizes MCTs into high- and low-grade, which may be helpful in predicting outcome and deciding whether to treat with adjuvant chemotherapy.12
4. Lung Tumor
Many canine lung tumors may be identified incidentally when taking thoracic radiographs for other presentations or signs. When staging for another type of cancer and a solitary lung mass is diagnosed, it is likely that this is a primary lung tumor rather than metastasis. These can occur in any lung lobe but are often seen in the peripheral caudal lung lobes.
Most solitary lung masses are carcinomas.13 Thoracic CT scan is recommended for staging to assess for lung metastasis and lymphadenomegaly.14 Evidence of either intrapulmonary metastasis or lymph node metastasis can significantly change prognosis with evidence of gross metastatic disease. A lung lobectomy can be performed via a lateral thoracotomy, median sternotomy, or minimally invasive thoracoscopy (Figure 4).


(A) Lateral thoracotomy for removal of a pulmonary bronchogenic carcinoma (located between the thumb and index finger of the surgeon’s right hand). (B) Resection of the mass with the aid of a thoracoabdominal stapling device
Biopsy of the hilar lymph nodes is recommended as part of staging. In dogs without evidence of metastasis and few features of malignancy on histopathology, the prognosis with surgery is excellent, and chemotherapy may not be recommended. If the mass is large, if there are lymph node metastases, or if there are a lot of histological features of malignancy, chemotherapy may be recommended, and the long-term prognosis is not as good.13
5. Splenic Mass
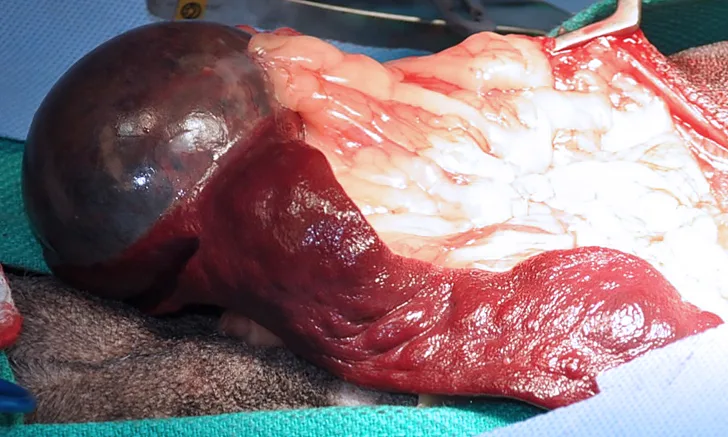
Splenic masses are commonly treated with splenectomy. In most cases, the tumor type is not known at the time of surgery. In the absence of a hemoabdomen, an ultrasound-guided fine-needle aspirate can be performed to determine tumor type of a solid mass. In cases of cavitated masses, with or without hemoabdomen, aspiration is a low-yield, potentially dangerous test and is not recommended. In the author’s opinion, if a small (<2-cm), noncavitated splenic mass is found as an incidental finding on abdominal ultrasound, close monitoring may be appropriate. Ultrasound should be repeated in a month, and splenectomy should be recommended if the mass is progressive. In most other cases, splenectomy to prevent rupture of the mass and hemoabdomen is recommended. If the patient is presented with a hemoabdomen, the splenectomy is an emergency (Figure 5). Staging should be performed with a minimum of 3-view thoracic radiographs and abdominal ultrasound (or thoracic and abdominal CT scan).

Splenic mass without hemoabdomen
Hemangiosarcoma is the most common malignant tumor of the spleen,15 but it is important to remember that not all splenic tumors are malignant. If the final diagnosis is hemangiosarcoma, the long-term prognosis for survival is poor, although adjuvant therapy may prolong survival.15