Corticosteroid Therapy in Veterinary Medicine
Elizabeth Rozanski, DVM, DACVECC, DACVIM, Cummings School of Veterinary Medicine at Tufts University
In modern veterinary medicine, glucocorticoids play a vital role in the management of an array of clinical conditions; however, they are associated with a variety of complications.
Steroids are widely used to treat inflammatory, immune-mediated, and neoplastic conditions. Steroids as a class include both mineralocorticoids and glucocorticoids.
Types of Steroids
Mineralocorticoids are involved in regulation of sodium and potassium through stimulation of sodium retention and promotion of potassium excretion. Aldosterone is the major naturally occurring mineralocorticoid.
Glucocorticoids are involved primarily in metabolism and immune-modulation, although a number of lesser functions have also been appreciated. Cortisol is the major naturally occurring glucocorticoid. It is released from the adrenal gland periodically, and larger amounts are released in times of stress.
The term steroid is commonly used by lay individuals to refer to anabolic steroids, which are used to improve athletic performance. Clinicians should be mindful that clients may be familiar with the term only as it applies to sports.
Related Article: Inhaled Corticosteroids & Airway Disease
Dosing Regimens
Synthetically manufactured steroids exhibit varying amounts of glucocorticoid and mineralocorticoid activity, which can make it difficult to determine how to dose a specific agent and what side effects might occur. The agents may also have different potency, meaning that on a milligram basis differing degrees of activity and duration of action are anticipated.
Steroid Selection
When choosing a steroid, keep in mind that most oral dosing regimens are provided as prednisone (prednisolone) equivalents. If prednisone is not available or is not tolerated a different drug, typically dexamethasone, can be substituted. Dexamethasone is 5 to 7 times more potent per milligram as prednisone (eg, 30 mg prednisone equals 4.3–6 mg dexamethasone). It is also available as sodium phosphate (dexamethasone-SP) which has a label concentration of 4 mg/mL; this is equivalent to 3 mg/mL of dexamethasone. It is more soluble than dexamethasone and has a more rapid onset of action.
For other injectable glucocorticoids, specific uses have been suggested or experimentally validated in humans, such as methylprednisolone sodium succinate for spinal cord trauma, although the actual relevance to veterinary patients remains to be determined.
With the exceptions of Addison’s disease, asthma, and severe allergic reactions, glucocorticoids have fallen out of favor in the emergency setting because multiple studies have not proven their use to be beneficial.
Types of Complications
Complications of glucocorticoid administration are predictable and primarily reflect the development of iatrogenic Cushing’s disease. If administered rapidly intravenously, glucocorticoids may cause vomiting. This can be prevented by administering more slowly or in combination with crystalloid fluids.
Classic complications of dogs undergoing glucocorticoid therapy include massive polyuria and polydipsia, hepatomegaly, thin skin, panting, and polyphagia. Hair regrowth is diminished, and ultimately, bilaterally symmetric alopecia may develop.
Other complications may include:
Steroid hepatopathy, predictable in dogs although rare in cats
Elevated liver enzymes, including alkaline phosphatase
Histopathologic hepatic changes
Marked lethargy
Panting (thought to result from hepatomegaly and skeletal muscle weakness)
Gastrointestinal signs, including gastric ulcerations
Diabetes mellitus or diabetic ketoacidosis
Pulmonary thromboembolism (Figure 1)
Urinary tract infections, including emphysematous cystitis infections
Pyometra in intact female dogs
Severe aggression (rare)

Pulmonary thromboembolism in a dog treated with immunosuppressive doses of prednisone for severe polyarthropathy
Exogenous steroids may block some clinical signs of infection or sepsis, such as fever, which may make detection of infection more challenging. Glucocorticoid therapy may be a risk factor for pancreatitis, although this is not universally accepted. Most side effects are more pronounced at higher doses.
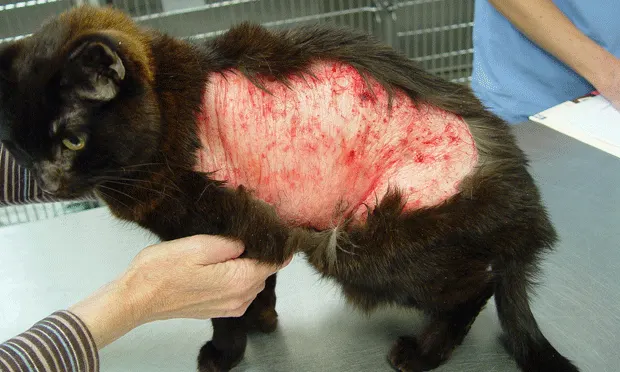
Cats are considered more resistant to the side effects of glucocorticoids than dogs, but may develop diabetes or iatrogenic Cushing’s disease (Figure 2).

Iatrogenic Cushing’s disease in a cat after long-term therapy with glucocorticoids
Preventing Complications
The best way to prevent complications is to avoid using steroids when possible. As with all diseases, therapy should be tailored to the individual patient, with other medications such as azathioprine or inhaled steroids such as fluticasone considered if indicated.
Excellent communication regarding patients that will be on long-term, high-dose glucocorticoids can improve client tolerance for side effects. Clients should be strongly counseled to avoid overfeeding, because obesity will magnify some side effects and may even affect survival. Pulmonary thromboembolism is the most devastating complication in patients with critical disease undergoing steroid therapy (eg, immune-mediated hemolytic anemia). Strong consideration should be given to simultaneous anticoagulation with either a low molecular weight heparin (eg, dalteparin, 150 IU SC Q 8–12 H) or ultralow–dose aspirin (0.5 mg/kg Q 12–24 H).
Treating Complications
Complications may require specific therapy, although, as always, steroids should be withdrawn or tapered if medically tolerable. The clinician should recall that the hypothalmic–pituitary–adrenal axis will have been suppressed by long-term administration of glucocorticoids and steroids should not be stopped suddenly. The length of the taper depends on the duration and extent of treatment. Short courses of glucocorticoids (1 week or less) need not be tapered before discontinuation.
Gastric Ulceration
Gastric ulceration induced by steroid therapy alone is uncommon, although when it occurs, bleeding may be impressive. Treatment of gastric ulceration includes use of proton-pump inhibitors (omeprazole), H-2 antagonists (famotidine), and sulcralfate. There is minimal evidence that concurrent therapy with H-2 antagonists is warranted during glucocorticoid therapy.
Diabetic Ketoacidosis & Diabetes Mellitus
Pets receiving glucocorticoids are prone to development of diabetic ketoacidosis and should be carefully evaluated if presented for vomiting or lethargy. Some cats, and very rarely dogs, will transiently become diabetic while undergoing steroid therapy. If persistent hyperglycemia and glucosuria develop, insulin therapy should be instituted, and glucocorticoids tapered if possible.
Diabetic patients receiving exogenous glucocorticoids will often be relatively insulin-resistant, although treatment is still successful. Diabetes mellitus, whether spontaneous or secondary to glucocorticoid therapy, typically requires exogenous insulin therapy. For pets with suspected glucocorticoid-induced diabetes, diabetic remission is possible following steroid withdrawal.
Infections
Infections may occur in a patient receiving steroid therapy, including deep and superficial pyodermas, urinary tract infections, and pyometra in intact bitches. Antibiotic therapy is advised, ideally guided by culture and sensitivity testing.
The newly diabetic pet receiving glucocorticoids should be assumed to have a urinary tract infection and be treated accordingly until disproved by urine culture results.
Cushing’s Disease
The classic Cushing’s appearance and clinical picture will resolve spontaneously as the glucocorticoids are tapered, although some dogs appear more sensitive than others. At the typically immunosuppressive dose of 2 to 4 mg/kg/day of prednisone, almost all dogs will show signs compatible with Cushing’s disease. In most dogs, resolution or marked improvement occurs at approximately 0.5 to 1 mg/kg/day, although signs are still often apparent. Excessive weight gain will lead to a longer duration of some signs (panting and exercise intolerance).
Editor's note: This article was originally published in March 2010 as "Steroid Therapy"