Feline Allergic Dermatitis (Feline Atopic Syndrome)
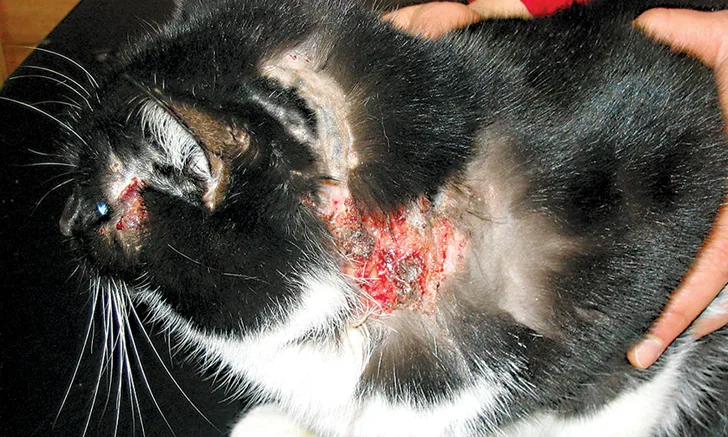
BEFORE TREATMENT A cat with feline atopic syndrome. Note the lesions and severe excoriations of the head and neck.
Brought to you by Elanco
Key Points
Do not minimize the importance of a thorough history and physical examination.
It is more effective in the pruritic patient to treat the disease, not just the pruritus.
Consider a trial with Atopica™ for Cats (cyclosporine oral solution) USP MODIFIED (Elanco) in cases of feline atopic syndrome.
The International Committee on Allergic Diseases of Animals has considered terminology for allergic skin diseases of cats and the consensus is to use feline atopic syndrome (FAS).1 This term specifically excludes parasitic causes, but includes environmental allergens and, because such reactions may occur together with atopic dermatitis in an individual cat, some manifestations of food reactions.
Pruritic skin diseases in cats can be broken down into 4 main categories: hypersensitivity (allergy), ectoparasitism, infection, and miscellaneous causes, including neoplasia and contact dermatitis.
Approach to the Cat with FAS
In the past, steroids—especially long-acting injections—were often used to control pruritus. While steroids may be appropriate for short-term alleviation of pruritus, long-term approaches revolve around use of Atopica for Cats or allergen-specific immunotherapy.
The best strategy derives from a thorough patient history, which is paramount in working up a pruritic patient. Using a dermatologic history form helps save time and ensures that important factors (age of onset, duration of pruritus, seasonality, other pets) are addressed. A good physical examination is equally important. Areas affected in cats vary slightly from those in dogs and may include the head, neck, ears, abdomen, caudal thighs, forelegs, and lateral thorax.
A 2012 report described the establishment of diagnostic criteria for FAS, at that time referred to as non-flea induced hypersensitivity disorder.2 The authors found that affected cats often demonstrate one of the following reaction patterns: pruritus of the head, neck, pinnae; miliary dermatitis; self-induced alopecia; or eosinophilic granuloma complex.2,3 Unfortunately, the type of pattern or lesion does not help differentiate the specific cause of the hypersensitivity in cats (ie, food vs flea vs environmental).
Once a history and physical/dermatological examination have been completed, the clinician should consider a few simple tests such as skin scraping for parasites and cytology for detecting yeast or bacterial overgrowth. The importance of flea allergy dermatitis should not be minimized. A 4- to 6-week therapeutic trial using a monthly product labeled for use in cats should be conducted in all cats not currently receiving flea control.
In cats with non-seasonal pruritus, a restricted diet trial should be conducted. Food allergens can cause flares of clinical signs of FAS. The gold standard is to use a restricted novel protein and/or hydrolyzed protein diet for 6 to 8 weeks, followed by provocation with the original food. Selection of the testing protein should be based on foods to which the cat has not been exposed. In multicat households, all cats should receive the hypoallergenic diet to prevent the trial cat from being exposed to food allergens.
Treatment
Bacterial infections are seen less often in cats than in dogs but can still play a role in feline pruritus. Infections can be either the cause of or secondary to pruritus and may involve the ears.
It is more effective in the pruritic patient to treat the disease, not just the pruritus. If, for example, a bacterial infection or parasite is present, treat it. If flea allergy, food allergy, and infection have been eliminated, then it is time to investigate FAS as a possibility.

AFTER TREATMENT WITH ATOPICA™ FOR CATS The cat shown on above after treatment with Atopica for Cats.
Cyclosporine has been documented in numerous clinical trials to be effective in the treatment of atopic dermatitis in dogs and is also used to treat feline allergic skin disease.4-6 The beneficial effects in atopy result from inhibition of T-cell activation, resulting in a decrease in pro-inflammatory cytokines such as IL-2.4 Suggested dosage is 7 mg/kg/d in cats, with reduction of the dosage when clinical signs have resolved.5,6
In a field study in which efficacy was assessed in 181 client-owned cats, 78.6% of cats treated with Atopica for Cats were graded as successes, compared with 26.2% of placebo-treated cats.5,6 Atopica for Cats can be a very affordable option in cats.7 The nephrotoxicity and hepatotoxicity concerns that have been seen in humans have not been noted as frequent/common concerns in animals. (Renal enzyme changes and weight loss resulting in hepatic lipidosis are noted on product label for Atopica for Cats.)5,8
Allergen-specific immunotherapy (ASIT) is considered safe and effective for treatment of FAS. Reported success in uncontrolled studies varies from 50% to 100%.9 Because intradermal allergy testing is difficult to perform and interpret in cats, the selection of allergens for ASIT is often determined by serum testing of IgE using an enzyme-linked immunosorbent assay. Some owners may find hyposensitization injections workable and others may prefer the ease of oral treatment with Atopica for Cats.
Some of the same strategies (bathing, epidermal barrier repair) used for atopic dogs can be applied to cats, but cats often are more resistant to the idea of frequent bathing. The use of sprays, wipes, or mousses may be preferable. Fatty acids can be given as an oral capsule or in a form added to the food (liquid or capsules opened onto the food). In addition, topical lipids or phytosphingosine can be applied to help strengthen the epidermal barrier.
ATOPICA™ for Cats (cyclosporine oral solution) USP MODIFIED is indicated for the control of feline allergic dermatitis as manifested by excoriations (including facial and neck), miliary dermatitis, eosinophilic plaques, and self-induced alopecia in cats at least 6 months of age and at least 3 lbs in body weight.
IMPORTANT SAFETY INFORMATION ATOPICA FOR CATS (cyclosporine oral solution) USP MODIFIED: Do not use in cats with a history or suspicion of malignant disorders, feline leukemia virus (FeLV) or feline immunodeficiency virus (FIV) infection, or hypersensitivity to cyclosporine. Atopica is a systemic immunosuppressant that may increase susceptibility to infection, development of neoplasia, and decrease response to vaccination. Persistent, progressive weight loss may result in hepatic lipidosis; monitoring of body weight is recommended. For use only in cats. Wash hands after administration. People with known hypersensitivity should avoid contact with Atopica. Do not use with other immunosuppressive agents. It is important for cats to avoid exposure to Toxoplasma gondii during treatment. Use with caution in cats with diabetes mellitus or renal insufficiency, and with drugs that affect the P-450 enzyme system. The most common adverse events were vomiting, weight loss, diarrhea or loss of appetite.
For full product and prescribing information, click here.
Atopica, Elanco and the diagonal bar logo are trademarks of Eli Lilly and Company or its affiliates. © 2017 Eli Lilly and Company or its affiliates. USCAFATO00070
