Canine Infectious Respiratory Disease Complex
Jonathan Dear, DVM, MAS, DACVIM (SAIM), University of California, Davis
Background & Pathophysiology
Canine infectious respiratory disease complex (CIRDC) is complex and multifactorial. Host, pathogens, and environment contribute to development of respiratory disease, which is primarily characterized by cough. In most instances, viral infection results in injury to the respiratory epithelium, which increases susceptibility to infection with bacteria, Mycoplasma spp, or other organisms that compound damage to the respiratory tract and contribute to progressive clinical signs.1
The disease has been referred to as kennel cough and infectious tracheobronchitis, but CIRDC is now preferred, as this term highlights both the involvement of multiple infectious agents and that disease susceptibility depends on host immunity and environmental factors. Moreover, although the term kennel cough may be useful in discussion with pet owners, it has become somewhat synonymous with Bordetella bronchiseptica and can cause confusion because dogs with appropriate vaccination history and robust immunity can still become infected with other CIRDC pathogens. Further, clinicians and owners should understand that antibacterial drugs are not effective when CIRDC is caused by a virus.
Although B bronchiseptica was the most commonly isolated bacterium in some previous studies,2-4 recent research has suggested numerous organisms likely play a role in CIRDC.5 Molecular techniques and virus isolation have identified mycoplasmal organisms,6 previously recognized viruses known to cause disease (eg, canine adenovirus-2, canine herpesvirus-1,7 canine parainfluenza virus,4 canine distemper virus [less frequent], canine respiratory coronavirus,8 various canine influenza viruses9), and emerging and novel viruses in which the link to clinical disease is not well-recognized (eg, canine reovirus,10 canine bocavirus, canine hepacivirus, canine pneumovirus11).
CIRDC has been documented worldwide, and no host-susceptibility–related genetic factors have been identified. Seasonal patterns of CIRDC have been observed; B bronchiseptica infections have been more commonly identified in dogs during cold months.5 Any dog can be infected with CIRDC-associated pathogens; however, young dogs and those with pre-existing airway disease (eg, bronchiectasis, airway collapse, ciliary dyskinesia) may be more susceptible due to impaired airway clearance. In addition, dogs in high-density environments12 or those that are immunologically naive or immunocompromised (endogenous or exogenous) are at increased risk and appear to develop more severe disease.13
History
In dogs presented with signs of upper respiratory tract disease, it is important to determine travel history and exposure to densely housed dog populations. Certain diseases (eg, canine influenza) have historically appeared to be geographically restricted, and travel to or contact with animals from these locations can raise suspicion for the primary pathogen. Similarly, a clue to the infectious pathogen may be related to the patient being recently boarded at a kennel (see Bordetella Bronchiseptica Pneumonia Treated with Glucocorticoids due to Chronic Cough), shelter, or veterinary hospital, as well as exposure to recent addition of animals to the household.14,15
Immunization history does not rule out CIRDC, but patients with up-to-date vaccinations against B bronchiseptica and parainfluenza virus appear to have attenuated clinical signs. Vaccines are not available for many viruses implicated in CIRDC.
Bordetella bronchiseptica pneumonia treated with glucocorticoids due to chronic cough
An 11-year-old intact female Pomeranian was presented for collapse refractory to previous therapy. She and her housemate had developed an acute cough after being boarded at a kennel. The housemate recovered without treatment. The patient had previously been given amoxicillin, enrofloxacin, prednisone, acepromazine, and hydrocodone to treat her cough, but no appreciable improvement was observed.
On examination, the dog was panting with expiratory effort and crackles auscultated diffusely. She exhibited an occasional goose-honking cough and retch. Her body temperature was normal. Radiography (Figures 1 and 2), fluoroscopy (Figure 3), and bronchoscopy (Figures 4 and 5) were performed.
Bronchoscopy revealed moderate grade 2 (40%-50%) extra-thoracic airway collapse, with grade 4 (100%) collapse of the intrathoracic airway (most severe at the carina). Marked diffuse purulent discharge was observed throughout the trachea and mainstem bronchi along with grade 3 to 4 (75%-100%) collapse of proximal bronchi (left cranial, right cranial, and right middle), with thickened mucosa and blunted airway divisions throughout. The caudal airways (left and right) were less affected distally.
Bronchoalveolar lavage samples revealed marked septic suppurative inflammation. The patient was initially treated with nebulization and empiric enrofloxacin (10 mg/kg PO once daily for 3 weeks) and amoxicillin/clavulanic acid (13.5 mg/kg PO twice daily for 3 weeks) pending culture and susceptibility results. Both B bronchiseptica and Mycoplasma spp were cultured from the bronchoalveolar lavage samples. B bronchiseptica was susceptible to most antimicrobials; a susceptibility panel was not performed for Mycoplasma spp. A recheck 2 weeks later revealed improved but persistent disease; the patient was subsequently transitioned to doxycycline (5 mg/kg PO twice daily for 2 weeks) for possible clinical activity against Mycoplasma spp.17
Because of her concurrent airway collapse, the patient was at risk for chronic cough without lifestyle modification. This case illustrates the need for comprehensive evaluation of dogs with acute or chronic cough, as many therapies (eg, glucocorticoids, cough suppressants, airway stenting) can prolong or exacerbate disease.

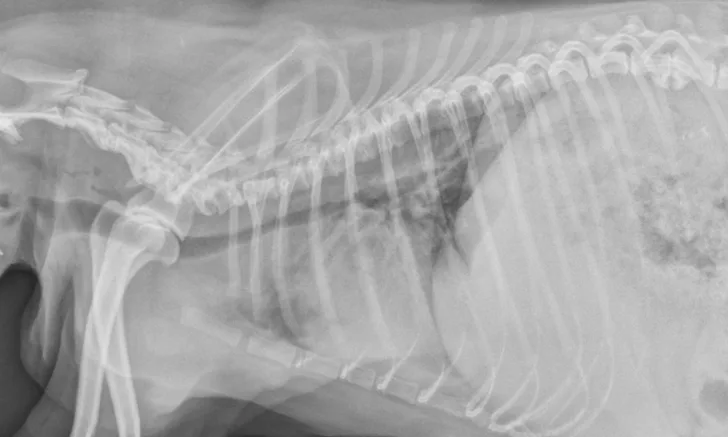
FIGURE 1
Left lateral radiograph showing dynamic collapse of the intrathoracic trachea from the level of the thoracic inlet to the mainstem bronchi. A small amount of esophageal gas is observed (asterisk), and the lungs are hypoinflated, which is likely secondary to increased respiratory effort. Mild, generalized cardiomegaly and hepatomegaly are also evident in the thorax (arrows) and abdomen (arrowheads), respectively.
Clinical Signs
Clinical signs of CIRDC are generally based on the degree of respiratory tract involvement, severity of damage, patient age, and pathogen virulence. CIRDC can be characterized as uncomplicated or complicated, depending on the severity of clinical signs. Uncomplicated disease generally results in an acute, self-resolving cough that can be triggered by exercise or excitement or occur spontaneously; retching and expectoration of clear mucoid respiratory secretions typically follows, but this is not specific for CIRDC or any specific organism. Complicated disease can progress to pneumonia, which manifests as the aforementioned cough and is often accompanied by hyporexia, fever, lethargy, hemoptysis, and difficulty breathing.
Physical examination of dogs with uncomplicated disease can be unremarkable or may include the presence of an inducible cough on tracheal palpation; however, this is not specific for CIRDC and it is possible to induce a cough with tracheal palpation in dogs with other respiratory disease. In dogs with complicated disease, a low-grade fever (103°F-104°F [39.4°C-40°C]) is not uncommon, respiratory rate and/or effort might be increased, and increased bronchovesicular sounds or crackles may be auscultated.
Diagnosis
Uncomplicated Disease
Diagnostic examination in dogs with uncomplicated disease can be unrewarding and unnecessary, as CBC, thoracic radiography, and arterial blood oxygen concentrations are generally within normal limits. Diagnosis is accomplished via a combination of compatible history and clinical signs, physical examination, and spontaneous resolution of disease.
Complicated Disease
In dogs with pneumonia as a result of complicated disease, CBC can reveal neutrophilic leukocytosis with a left shift; thoracic radiographs may show an interstitial to alveolar pattern with cranioventral distribution (characteristic of bacterial pneumonia), diffuse interstitial pneumonia (observed with viral disease), or a mixed pattern; and radiographs can help investigate differential diagnoses (eg, fungal pneumonia, pyothorax, congestive heart failure).
Ideally, airway samples from dogs with complicated disease should be submitted for cytologic evaluation and culture and susceptibility testing (aerobic and mycoplasmal culture). PCR can be performed on airway washes when specific viral diseases (ie, distemper, influenza, canine parainfluenza virus) are suspected, but caution should be used when interpreting respiratory PCR panels because the sensitivity and specificity of these tests are unknown.16
Treatment & Management
Uncomplicated Disease
Outpatient therapy and isolation from other dogs for 2 to 3 weeks is recommended to minimize the risk for transmission. Therapy should include adequate nutrition, hydration, and hygiene. Cough suppressants (eg, hydrocodone [0.22 mg/kg PO every 8-12 hours] or butorphanol [0.1-0.4 mg/kg PO every 8-12 hours]) can be used when bacterial pneumonia is unlikely. Antimicrobial therapy is not typically needed, but doxycycline (5 mg/kg PO every 12 hours for 7-10 days) can be administered if disease persists >10 days or as a first-line antimicrobial if clinical signs progress to systemic signs (ie, fever, lethargy, decreased appetite).17
Setting Realistic Client Expectations Is an Essential Part of Treatment
Most patients with CIRDC respond well to supportive care and adequate time to recover, but helping clients understand this can be challenging. Although clients may appreciate the lower costs associated with limited testing and treatment, they often expect specific therapies and may react negatively to a do-nothing approach without adequate explanation.
One of the most common expectations clients may have is that their dog will go home with antimicrobials. It is important to acknowledge their concerns and make sure they know that follow up, including antibiotics if necessary, is part of the plan.
Sample conversation: Client: Doesn't Biscuit need antibiotics? I don't want him to get worse.
Clinician: Antibiotics are sometimes used but are not needed in this case. We reach for antibiotics when dogs have more severe disease. Antibiotics can cause complications like vomiting and diarrhea, so we would rather not give them unless absolutely necessary. Biscuit will need antibiotics if he develops pneumonia, which is unlikely but should be monitored for. If he loses his appetite, has less energy, starts breathing harder, or has a worsened cough, we will consider antibiotics. Right now, he is better off without them, and it is easier on him and you because you will not have to administer pills.
For more CIRDC communication tips and sample conversations, read the full article by J. Scott Weese, DVM, DVSc, DACVIM, FCAHS, at https://www.cliniciansbrief.com/article/cirdc-setting-client-expectations.
Complicated Disease
Inpatient therapy should be considered for dogs with bacterial pneumonia if hydration would be inadequate with outpatient therapy or if oxygen therapy is necessary. In these dogs, IV fluid therapy and parenteral antibiotics are generally warranted; empiric antimicrobial therapy with either doxycycline (5 mg/kg IV every 12 hours) or a combination of clindamycin (5-10 mg/kg IV every 8 hours) or ampicillin (10-20 mg/kg IV every 8 hours) and a fluoroquinolone (ie, enrofloxacin [10 mg/kg IV every 24 hours]) can be considered. At discharge, enteral therapy can be continued with doxycycline (5 mg/kg PO every 12 hours) or amoxicillin/clavulanate (12-20 mg/kg PO every 12 hours), ideally, based on culture and susceptibility testing.17 These patients should be strictly isolated from other patients, and veterinary staff should use personal protective equipment and proper hygiene to prevent transmission via fomites. Patients with significant respiratory compromise should be provided with supplemental oxygen in an oxygen cage (if available) or through a nasal cannula.
Bronchodilators (eg, theophylline [5-10 mg/kg PO every 12 hours], terbutaline [0.625-5 mg/dog SC every 8-12 hours]) are rarely indicated in dogs with CIRDC but can be administered in patients suspected to have bronchospasm that is impairing appropriate ventilation. If used concurrently with medications metabolized by cytochrome P450 enzyme system, the dosage should be altered. In the case of fluoroquinolones, the theophylline dose should be reduced due to drug interactions that can lead to reduced metabolism and subsequent increase in plasma levels, which might result in adverse effects.
Prognosis & Prevention
Complete resolution of uncomplicated disease typically occurs within 10 to 14 days after onset of clinical signs. Complicated disease can require treatment for 2 to 6 weeks, and, although prognosis is generally good, subsequent development of severe pneumonia can progress to sepsis or multiple organ dysfunction due to systemic inflammatory response syndrome.
According to AAHA vaccination guidelines,18 immunization is against core (eg, canine distemper virus, canine adenovirus-2) and noncore (eg, B bronchiseptica, canine influenza virus, canine parainfluenza virus) CIRDC pathogens. When administered appropriately, core vaccines provide robust immunity against these viruses. Immunization with noncore vaccines may not prevent infection but does appear to limit the severity of clinical signs and duration of shedding postinfection. Oral, intranasal, and parenteral B bronchiseptica (modified-live and killed) vaccines are commercially available. In general, parenteral vaccines produce a profound serologic response, but oral and intranasal vaccines appear to provide more effective mucosal immunity, with intranasal vaccines resulting in less severe disease in dogs challenged with infection in experimental studies19,20; thus, intranasal vaccination has been shown to produce protective immunity for at least 1 year. To the author’s knowledge, these studies have not been published for oral or parenteral B bronchiseptica vaccines. Vaccines are available as monovalent products or in combination with other respiratory pathogens (eg, canine parainfluenza virus, canine adenovirus-2). Vaccines are available for 2 strains of canine influenza (ie, H3N2, H3N8) as both monovalent and bivalent formulations. As killed vaccines, they induce a less robust immune response than avirulent live viruses and are likely to reduce severity of disease rather than prevent infection.
Clinical Follow-up/Monitoring
Recheck of dogs with uncomplicated disease is often unnecessary. In dogs with suspected bacterial pneumonia as a result of complicated disease, radiography should ideally be performed every 2 to 3 weeks while monitoring for resolution of clinical signs. Previously, antimicrobials were administered for 3 to 6 weeks or at least 1 to 2 weeks beyond resolution of clinical and/or radiographic signs of disease. The International Society for Companion Animal Infectious Diseases guidelines suggest that a shorter duration may be adequate; thus, the decision to continue antimicrobials should be made on a case-by-case basis using clinical, hematologic, and radiographic findings to guide treatment.17
Following onset of signs, dogs should be isolated for 2 to 3 weeks, and care should be taken to avoid sharing of bedding, bowls, and other husbandry items to prevent transmission to susceptible dogs.
Conclusion
CIRDC is a common cause of acute cough in dogs, especially those cohoused with or exposed to other dogs (eg, shelters, dog shows, grooming facilities). Generally, this condition is self-limiting with supportive care, but some dogs need more advanced intervention, including hospitalization or antimicrobial therapy if bacterial or mycoplasmal pneumonia develop.